kbg00 | ACSJCA | JCA11.2.5208/W Library-x64 | manuscript.3f (R5.2.i3:5013 | 2.1) 2022/08/03 13:05:00 | PROD-WS-121 | rq_629302 | 7/27/2023 16:24:08 | 20 | JCA-DEFAULT
![]()
![]()
kbg00 | ACSJCA | JCA11.2.5208/W Library-x64 | manuscript.3f (R5.2.i3:5013 | 2.1) 2022/08/03 13:05:00 | PROD-WS-121 | rq_629302 | 7/27/2023 16:24:08 | 20 | JCA-DEFAULT
![]()
![]()
pubs.acs.org/EF
Review
1 Advances in Catalytic Hydroconversion of Typical Heavy Carbon
2 Resources under Mild Conditions
3 Xian-Yong Wei,* Xiang Bai, Feng-Yun Ma, Zhi-Min Zong, Wei Zhao, Zhong-Hai Ni, Xing Fan,
4 Lin-Bing Sun, Jing-Pei Cao, Yun-Peng Zhao, Shi-Chao Qi, Jing Liang, Xiao-Ming Yue, Fang-Jing Liu,
5 Wen-Long Mo, Jing-Mei Liu, Yu-Hong Kang, Guang-Hui Liu, Zhong-Qiu Liu, and Li Li
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]() ACCESS Metrics & More Article Recommendations *sı Supporting Information
ACCESS Metrics & More Article Recommendations *sı Supporting Information
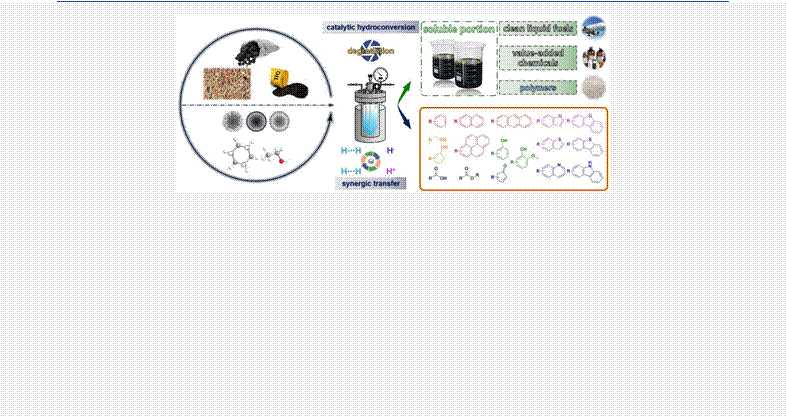 6 ABSTRACT: Low-rank coals, biomass, and heavy petroleum are typical heavy carbon resources, which are currently
not efficiently
6 ABSTRACT: Low-rank coals, biomass, and heavy petroleum are typical heavy carbon resources, which are currently
not efficiently
7 used, and many processes for the current use of the heavy carbon resources cause severe environmental pollution with very limited
8 economic benefits even with a great deficit. Efficiently using the heavy carbon resources can greatly improve the environment and
9 create huge economic benefits. The main difficulty in efficiently using the heavy carbon resources is the high content of aromatic
10 rings (ARs) and heteroatoms in addition to the insoluble and complex macromolecular structures of the heavy carbon resources.
11 Catalytic hydroconversion (CHC) includes catalytic hydrocracking and subsequent catalytic hydrofining. Many low-rank coals
12 consist of macromolecular species, which are rich in bridged linkages connecting structural units, especially ARs, on which side
13 chains are usually contained. Many ARs, bridged linkages, and side chains contain heteroatoms. Over a proper active catalyst, many
14 bridged linkages and even some side chains can be cleaved, and many heteroatoms outside the ARs can be removed by the CHC
15 under mild conditions to get soluble portions with relatively simple composition, facilitating subsequent separation to obtain pure
16 chemicals, especially value-added products, such as condensed aromatics. The remaining economically inseparable species can be
17 converted to liquid chemicals, especially nonsubstituted and alkyl-substituted cyclanes, by subsequent catalytic hydrofining. Catalysts
18 and catalytically formed active hydrogen play crucial roles in the processes. Mainly based on our investigations, the related advances
19 are reviewed in this work.
20 Low-rank coals, biomass, and heavy petroleum are typical heavy
21 carbon resources.1−4 Such heavy carbon resources are currently
22 not efficiently used, and many processes, including catalytic
23 hydroconversion (CHC) at high temperatures, pyrolysis, and
24 gasification, for the current use of heavy carbon resources cause
25 environmental pollution, e.g., inevitable exhaust gas, wastewater,
26 and waste residue, to different extents, with very limited
27 economic benefits even with a great deficit.
28 Efficiently using heavy carbon resources can greatly improve
29 the environment and create huge economic benefits. Most of
30 heavy carbon resources have high contents of aromatic rings
31 (ARs), especially condensed ARs, and heteroatoms in addition
32 to the insoluble and complex macromolecular structures,
 causing huge difficulty in their efficient
use. When using heavy 33 carbon
resources as precursors for producing clean fuels, both 34
ARs and heteroatoms should
be removed, but as chemicals, 35
many aromatics, especially condensed and heteroatom-contain- 36 ing aromatics in heavy carbon resources and the soluble
portion 37 from converting the heavy carbon
resources, are valuable, and in 38 general, the aromatics with
more ARs and more heteroatoms are 39
causing huge difficulty in their efficient
use. When using heavy 33 carbon
resources as precursors for producing clean fuels, both 34
ARs and heteroatoms should
be removed, but as chemicals, 35
many aromatics, especially condensed and heteroatom-contain- 36 ing aromatics in heavy carbon resources and the soluble
portion 37 from converting the heavy carbon
resources, are valuable, and in 38 general, the aromatics with
more ARs and more heteroatoms are 39
![]() © XXXX American Chemical Society
© XXXX American Chemical Society
A
https://doi.org/10.1021/acs.energyfuels.3c01713
Energy Fuels XXXX, XXX, XXX−XXX
40 more expensive.5 For example, the price of triphenylene with
41 condensed 4 ARs is 962 times that of naphthalene with
42 condensed 2 ARs, and with condensed 3 ARs, the price of
43 acridine is 7.25 times that of anthracene.4
44 CHC includes catalytic hydrocracking and subsequent
45 catalytic hydrofining. Both low-rank coals and biomass,
46 especially their insoluble portions, mainly consist of macro-
47 molecular species, which are rich in bridged linkages connecting
48 structural units, especially ARs, on which side chains are usually
49 contained. Since soluble portions in heavy carbon resources
50 usually consist of numerous organic species, using an insoluble
51 portion as the reactant can prevent the disturbance of the
52 inherently existing organic species in the soluble portions.6−9
53 Thermal conversions at high temperatures are still the main
54 technologies and the main investigations for utilizing heavy
55 carbon resources. As mentioned above, such conversions
56 consume large amount of heavy carbon resources with the
57 huge emission of exhaust gas (especially CO2), wastewater, and
58 waste residue to produce low-value products. Developing
59 directional conversion technologies, especially CHC under
60 mild conditions, is crucial for smartly converting heavy carbon
61 resources to value-added products, especially condensed
62 aromatics, with near-zero emission of exhaust gas, wastewater,
63 and waste residue. Unfortunately, such important investigations
64 were paid less attention compared to thermal conversions. The
65 key scientific issues to be resolved for directional CHC of heavy
66 carbon resources are as follows: (1) How does one precisely
67 tailor the macromolecular species in the heavy carbon resources
68 to obtain value-added products? (2) What are active hydrogen
The first reason is that macromolecular species in some heavy 101 carbon resources contain weak bridged linkages and bond 102 scissions during the NCHC, e.g., coal pyrolysis, result solely 103 from the thermolysis of weak bridged linkages, so the hydrogen- 104 donating compounds donate their benzylic hydrogen to stabilize 105 radical fragments from the thermolysis of weak bridged 106 linkages,19 as illustrated in Scheme 1. However, the results 107 s1
![]()
Scheme 1. Conventional Consideration on the Role of Hydrogen-Donating Compounds in Promoting the Degradation of Heavy Carbon Resources by Stabilizing the Thermally Produced Radical Fragments19
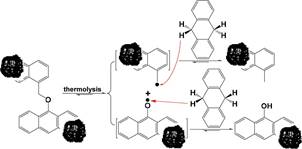
![]()
from the thermolysis of either 1,2-di(1-naphthyl) ethane or 1,3- 108 diphenylpropane indicate that adding a hydrogen-donating 109 compound inhibit each thermolysis and adding a hydrogen- 110 donating compound with a stronger hydrogen-donating ability 111
69 species for precisely tailoring the macromolecular species and
inhibits the thermolysis more severely.
For example, 1,3- 112
70 how does one effectively produce the active hydrogen species?
71 (3) Where are the key positions to be tailored in the
72 macromolecular species and what roles do the active hydrogen
73 species play in tailoring the positions? Mainly based on our
74 investigations, the related advances are reviewed in this paper in
75 comparison with noncatalytic hydroconversion (NCHC) of
76 heavy carbon resources.
diphenylpropane thermolysis is a typical chain reaction and 113
thermally the produced radical fragments, such as benzyl, 114 phenylethyl, and 1,3-diphenylprop-1-yl radicals, play crucial 115 roles in the chain reaction. Benzylic hydrogen provided by a 116 hydrogen-donating compound scavenges the radical fragments 117 and thereby inhibits 1,3-diphenylpropane thermolysis. These 118 facts suggest that scavenging radical fragments by a hydrogen- 119 donating compound could inhibit the NCHC of heavy carbon 120
resources.
121
78 Benzylic hydrogen exists in both heavy carbon resources and
79 hydrogen-donating compounds used as solvents. Hydrogen-
80 donating compounds are generally considered to promote the
81 conversion of heavy carbon resources, especially coal
82 liquefaction.10−15 Sheng et al.16 investigated NCHC of an
83 asphaltene using tetralin as a hydrogen-donating compound.
84 Their results showed that the total yield of soluble portion from
85 asphaltene conversion reached 70.34% by controlling the
86 reaction conditions. Unfortunately, they did not provide the
87 detailed analyses, especially detailed molecular composition, of
88 asphaltene and the liquid. In fact, since both asphaltene and the
89 liquid consist of complex organic species, and since tetralin and
90 its derivates17 could be included in the liquid, getting the
Another reason is that benzylic hydrogen leaving from a 122
hydrogen-donating compound can attack an ipso-position 123 connecting a arylmethyl group and subsequently cleave the 124 strong >Car−CH2− bond. Such a pathway was first proposed by 125 McMillen et al.22 according to their investigation on the kinetics 126 of diphenylmethane decomposition in tetralin. However, 127 diphenylmethane conversion is less than 0.1% even in 20 h 128 reaction at 400 °C and still negligible even in a stronger 129 hydrogen-donating compound according to their kinetic 130 investigation. In fact, NCHC of diphenylmethane is very 131
difficult in tetralin even at 430 °C under pressurized H2.23 132
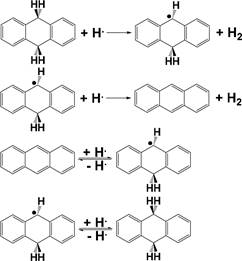
Taking 9,10-dihydroanthracene as an example, as depicted in 133 Scheme 2, thermally cleaving the >Cα-H bond in 9,10- 134 s2 dihydroanthracene to produce H• and 9-hydroanthr-10-yl 135
radical is relatively easy because of the relatively stable 9- 136
91 objective asphaltene conversion and total liquid yield is difficult.
92 Apparently, the hydrogen-donating ability of the hydrogen-
93 donating compounds increases in the order of tetralin <9,10-
94 dihydrophenanthrene <9,10-dihydroanthracene, since the
95 stability of the resulting radical fragments increases in the
96 same order, i.e., tetral-1-yl radical < 9-hydrophenanthr-10-yl
97 radical < 9-hydroanthr-10-yl radical.18 The promotional roles of
98 the hydrogen-donating compounds in the NCHC of heavy
99 carbon resources are related to two reasons rather than to any
100 hydrogen-donating compound.
Scheme 2. “Hydrogen Donation” from 9,10- Dihydroanthracene Leading to the Formation of Anthracene24
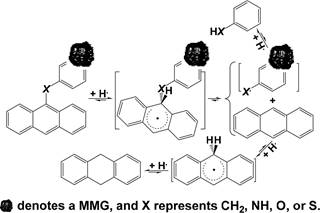
Scheme 3. An Example for Cleaving Bridged Linkages by Intramolecular Benzylic Hydrogen Transfer3
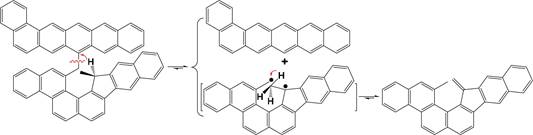
![]()
Scheme 4. Possible Pathway for the Selective Deoxygenation of Vanillin to 4-Methylguaiacol
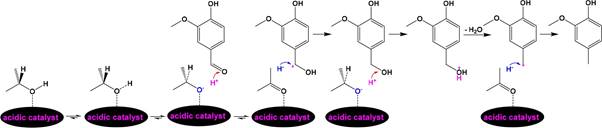
![]()
Scheme 5. A Chain Reaction from FeS2 Thermolysis to FeS2 Regeneration with H2 to Release H•
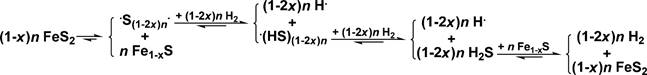
![]()
137 hydroanthr-10-yl radical, but the resulting H• prefers to abstract
138 benzylic hydrogen in 10-position of 9-hydroanthr-10-yl radical
139 due to the formation of much more stable anthracene rather
140 than being donated to an ipso-position in a diarylmethane. In
141 addition, such an intramolecular benzylic hydrogen transfer is
142 more favorable.
143 In some cases, intramolecular benzylic hydrogen transfer
144 could result in cleaving a macromolecular component to a
s3 145 smaller compound, as Scheme 3 illustrates. Such an intra-
146 molecular benzylic hydrogen transfer could proceed at relatively
147 low temperatures and is desirable for producing valuable
148 condensed arenes, but at temperatures higher than 480 °C,
149 the dehydrogenation from condensed arenes and subsequent
150 condensation among the condensed aromatic radicals to
151 produce chars are inevitable.
152 Alkanols, such as methanol, ethanol, propan-1-ol, and 2-
153 propanol, are also considered to be hydrogen-donating
154 compounds.25−31 However, since the dissociation energies of
was completely converted to ethanol-soluble portion and the 168 total yield of aromatics reached 25.5%. Over an acidic catalyst, 169 H+ transfer from 2-propanol to vanillin for selective deoxyge- 170 nation was reported to proceed under mild conditions without 171 H2.42 In this case, H+ could result from the adsorption of oxygen 172 atom in 2-propanol, which is an excellent nucleophilic 173 compound, and subsequent heterolytic cleavage of the −O−H 174 bond. The resulting (CH3)2CHO− adsorbed on the catalyst 175 tends to be converted to acetone by releasing H− from the 176 tertiary carbon. H+ transfer to the oxygen atom in the formyl 177 group of vanillin and subsequent H− abstraction by the benzylic 178
carbon lead to the formation of 4-hydroxymethylguaiacol 179 followed by H+ transfer to the oxygen atom in the −CH2OH 180 group of 4-hydroxymethylguaiacol, dehydration from the 181 protonated −CH2OH group, and H− abstraction by the 182 resulting guaiacylmethylium to produce 4-methylguaiacol 183 (Scheme 4). Noteworthily, transferring H+ to the oxygen atom 184 s4
155 − O−H bonds are much higher than those of ARCH2−H bonds,
156 directly donating H• by cleaving − O−H bonds is difficult. In
157 fact, the alkoxy group in an alkanol is a nucleophilic group, which
158 can attack a carbon atom connected with an oxygen atom and
159 subsequently cleave the >CH2−O− or >Car−O− bonds
in both 2-propanol and acetone also consumes H+.
185
186
187
160 followed by hydrogen transfer from the −OH group in the
161 alkanol to the oxygen atom; i.e., the hydrogen donation of
162 alkanols is induced by alkanolyses.32−40
163 Alkanols are usually used as solvents for the hydroconversion
164 of lignin and its derived monomers. Bai et al.41 examined NiMo/
165 Al-catalyzed hydroconversion of a corncob-derived residue in
166 ethanol under 2.76 MPa of initial hydrogen pressure (IHP) at
The roles of benzylic hydrogen in the CHC of heavy carbon 188
resources were intensively investigated. According to conven- 189 tional viewpoints,43−49 catalysts promote hydrogen transfer 190 from the hydrogen-donating compound, e.g., tetralin, to the 191 heavy carbon resource and from H2 to the dehydrogenated 192 hydrogen-donating compound, e.g., naphthalene, in addition to 193 the role of the hydrogen-donating compound in scavenging 194
167 320 °C for 7.5 h. Under such reaction conditions, the residue
radical fragments mentioned above.
195
196 Indeed, many hydrogen-donating compounds play positive
197 roles in swelling and dissolving organic matter in heavy carbon
198 resources, but their negative roles in the CHC of heavy carbon
with either a stronger hydrogen-donating ability or a more 236 condensed AR more severely scavenges H•. As a commonly used 237 hydrogen-donating compound, tetralin proved to apparently 238
199 resources cannot be ignored. Ouchi and Makabe50 first found
200 the inhibiting effect of tetralin in comparison with decalin, a non-
inhibit both di(1-naphthyl) methane hydrocracking over FeS2 and the reaction of coal macromolecule to oil.51
239
240
201 hydrogen-donating compound, on the CHC of bibenzyl and
202 benzyloxybenzene at 250 and 400 °C over stabilized nickel.
203 They considered that the inhibiting effect resulted from the
204 stronger adsorption of tetralin than that of decalin on the surface
205 of stabilized nickel and the main reaction proceeded by direct
206 hydrogen transfer from H2 over the surface of stabilized nickel.
207 Similar suggestion on the main reaction was also proposed by
208 Niu et al.51
In addition to the above defects, both hydroarenes used as 241
hydrogen-donating compounds and their resulting condensed 242 arenes are usually expensive and less volatile, suggesting that 243 they should be recovered as completely as possible after use, but 244 recovering them from a reaction mixture is very energy- and 245 time-consuming. Another problem is that since both hydro- 246 arenes used as hydrogen-donating compounds and their 247 resulting condensed arenes exist in organic matter in heavy 248
209 FeS2 proved to be active for producing H• by a chain reaction
carbon resources and their soluble products,
distinguishing 249
s5 210 shown in Scheme 5 without any hydrogen-donating com-
211 pound52 and H• plays a crucial role in cleaving the >Car−CH2−
their sources, i.e., whether they result from the hydrogen- 250
donating compounds or the heavy carbon resources, is difficult. 251
212 bridged linkages in diarylmethanes.53 However, FeS2-catalyzed
Some carbon materials, such as activated carbon
and 252
•
213 hydrocracking of diphenylmethane in decalin at 400 °C
carbon black,
also catalyze the formation and transfer of H
253
214 indicated that adding a hydrogen-donating compound, such as
215 tetralin, 9,10-dihydrophenanthrene, and 9,10-dihydroanthra-
216 cene, inhibited diphenylmethane hydrocracking and adding a
217 stronger hydrogen-donating compound inhibited diphenyl-
218 methane hydrocracking severer.24 Diphenylmethane hydro-
219 cracking was more inhibited by adding the hydrogen-donating
by homogeneously splitting H−H bond in H2. Adding sulfur 254
promotes di(1-naphthyl) methane hydrocracking over an 255 activated carbon due to the formation of H2S by the reaction 256 of H2 with the added sulfur over the activated carbon and the 257 much weaker H−S−H bond than H−H bond, and the 258 synergistically increased di(1-naphthyl) methane conversion 259
220 compound-derived condensed arenes, i.e., naphthalene, phe-
increases with raising the reaction temperature up to 350 °C.
260
221 nanthrene, and anthracene, and diphenylmethane conversion in
Previous investigation on the CHC of α,ω-diarylal- 261
222 the condensed arenes added decreased in the order of
kanes
indicated that the effectiveness of H•
transfer 262
223 naphthalene > phenanthrene > anthracene, being the same as
224 the order of the resulting hydrogen-donating compounds, i.e.,
for cleaving bridged linkages connecting ARs over FeS2 under 263
mild conditions not only depended on the H•-accepting ability, 264
225 tetralin >9,10-dihydrophenanthrene >9,10-dihydroanthracene,
in terms of superdelocalizability (Figure 1),
of an AR, but 265 f1
226 since the more condensed arenes more strongly adsorb on the
was also related to the stability, in terms of resonance energy 266
227 catalyst surface, and there are equilibriums between the
(Figure 2),
of the leaving arylalkyl or diarylalkyl radical.
267 f2
228 hydrogenation of condensed arene to its resulting hydrogen-
According to the CHC of α,ω-diarylalkanes at 300 °C over 268
FeS2,
only very small amount of biphenyl was converted to 269
229 donating compound, e.g., anthracene to 9,10-dihydroanthra-
230 cene, and the dehydrogenation of a hydrogen-donating
231 compound to its corresponding condensed arene, e.g., 9,10-
232 dihydroanthracene to anthracene, under pressurized H2 over
233 FeS2. Such an intramolecular hydrogenation and dehydrogen-
234 ation mainly consumes H• rather than donates H• to another
s6 235 molecule (Scheme 6). These results indicate that an additive
![]()
Scheme 6. Intramolecular H• Transfer between 9,10- Dihydroanthracene and Its Dehydrogenated Products4
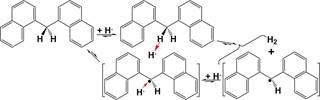
cyclohexylbenzene, while both bibenzyl and 1,3-diphenylpro- 270 pane were not converted due to the weak H•-accepting ability of 271 benzene ring (BR) and the resulting labile phenylethyl and 272 phenylpropyl radicals. Under the same conditions, 1,2-di(1- 273 naphthyl) ethane hydrocracking is not significant and the main 274 products are partially hydrogenated derivates due to the much 275 stronger H•-accepting ability of naphthalene ring (NR) than 276 that of BR but the still labile naphthylethyl radical. Bi(1- 277 naphthyl) cannot be directly hydrocracked under the same 278 conditions because of the resulting extremely labile phenyl 279 radical, but both naphthalene and tetralin were detected from 280 the CHC of bi(1-naphthyl) since H• transfer to the ipso-position 281 of a NR in bi(1-naphthyl) and subsequent H• transfer to the 282 same NR produced 1-(tetralyl) naphthalene, and subsequent H• 283 transfer to the ipso-position of NR in 1-(tetralyl) naphthalene 284
 produced naphthalene and tetralin.
produced naphthalene and tetralin.
285
Either iron sulfide- or nickel sulfide-catalyzed reaction of 9,10- 286 diphenylanthracene primarily proceeds by H• transfer to the 9- 287 and 10-positions of 9,10-diphenylanthracene, producing 288 (9R,10R)-9,10-diphenyl-9,10-dihydroanthracene as the main 289 product and (9R,10S)-9,10-diphenyl- 9,10-dihydroanthracene 290 as the byproduct71,72 because of the much larger super- 291 delocalizability of 9- and 10-positions than other positions 292 (Figure 1 and Scheme 7). H• can attack the 10-position of the 293 s7 intermediate 9,10-diphenyl-9-hydroanthryl radical via either the 294 same direction as the H• transfer to 9-position of 9,10- 295 diphenylanthracene or the opposite direction. In the case of 296 the H• transfer via the same direction, the added H• in the 9- 297 position tends to be abstracted by the adding H•, while in 298
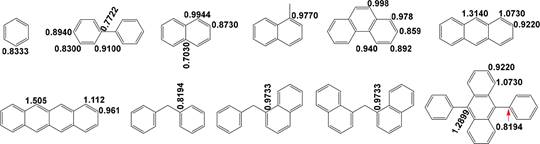
Figure 1. Superdelocalizabilities of carbon atoms in different positions of the typical arenes.21,69
![]()
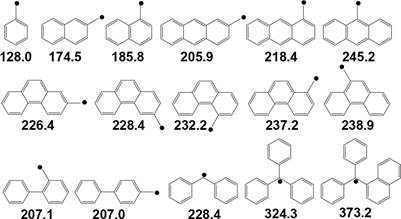
![]() Figure 2. Resonance energies
(kJ mol−1) of some benzylic
radicals.70
Figure 2. Resonance energies
(kJ mol−1) of some benzylic
radicals.70
Scheme 7. Reaction Pathway for H• Transfer to 9,10-
and (9R,10S)-9,10-diphenyl-9,10-dihydroanthracene (Scheme 312
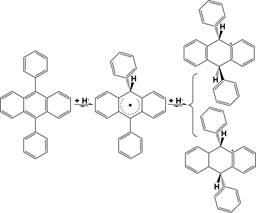 The CHC of 1-benzylnaphthalene at 300 °C over
FeS2 314 predominantly proceeds via H• transfer to the ipso-position of 315 NR in 1-benzylnaphthalene also due to the
much stronger H•- 316 accepting ability
of NR than BR.53 However,
2-benzylnaph- 317
The CHC of 1-benzylnaphthalene at 300 °C over
FeS2 314 predominantly proceeds via H• transfer to the ipso-position of 315 NR in 1-benzylnaphthalene also due to the
much stronger H•- 316 accepting ability
of NR than BR.53 However,
2-benzylnaph- 317
thalene and two 2-benzyltetralins were unexpectedly detected. 318 Their formation should be ascribed to the addition of the 319 relatively reactive benzyl radical, the leaving radical from the NR 320 via NR-CH2BR bond cleavage, to the 2-position of the NR. 321 Similar to FeS2, activated carbon predominantly catalyzes the 322 partial hydrogenation of condensed arenes. The reactivities of 323 condensed arenes toward the partial hydrogenation were found 324 to be closely related to the H•-accepting abilities of the 325
![]()
299 another case the adding H• cannot abstract the added H• so that
300 the transhydrogenation to 9,10-diphenylanthracene is predom-
The above results suggest that cleaving bridged linkages in 327 heavy carbon resources by H• transfer under mild conditions 328 over both metal sulfides and carbon materials is selective to 329 some extent. Using a tetrahydrofuran/methanol mixture− 330 insoluble portion of Pingshuo bituminous coal as an example, 331 its CHC over an iron−sulfur system and NCHC at 300 °C in 332
301 inant. H• transfer to either (9R,10R)-9,10-diphenyl-9,10-
cyclohexane were investigated.
As a result, the CHC produced 333
302 dihydroanthracene or (9R,10S)-9,10-diphenyl-9,10-dihydroan-
303 thracene is difficult due to the small superdelocalizability of the
304 BRs in (9R,10R)-9,10-diphenyl-9,10-dihydroanthracene and
305 (9R,10S)- 9,10-diphenyl-9,10-dihydroanthracene. In particular,
306 H• transfer to the *C in (9R,10R)-9,10-diphenyl- 9,10-
307 dihydroanthracene and (9R,10S)-9,10-diphenyl-9,10-dihy-
308 droanthracene is the most difficult, since the superdelocaliz-
309 ability of *C is smallest and the transferring H• can be easily
310 abstracted by the benzylic hydrogen, which is also a tertiary
311 hydrogen, in (9R,10R)-9,10-diphenyl-9,10- dihydroanthracene
much more anthracene and 9,10-dihydroanthracene than the 334
NCHC. In addition, the yield of 9,10-dihydroanthracene is 335 much lower than that of anthracene from the NCHC, while the 336 yield of 9,10-dihydroanthracene is appreciably higher than that 337 of anthracene from the CHC. The insoluble portion should 338 contain or even rich in anthracene ring, and some ARs could be 339 connected with a macromolecular group (MMG)-substituted 340
BRCH2-, BRO-, BRS-, and/or BRNH- group. H• transfer to the 341
ipso-position in the anthracene ring induced the cleavage of the 342
MMG-substituted BRCH2-anthracene ring, BRO-anthracene 343
344 ring, BRS-anthracene ring, and/or BRNH-anthracene ring, and
345 thereby released anthracene, which can be easily converted to
s8 346 9,10-dihydroanthracene by subsequent H• transfer (Scheme 8).
![]()
Scheme 8. Possible Pathway for the Release of Anthracene and 9,10-Dihydroanthracene from the CHC of a
1,2,3,4,5,6,7,8-octahydroanthracene as the byproducts under 5 371 MPa of IHP at 150 °C over Ni/ZSM-5 for 5 h.77 Interestingly, all 372 the products kept the central BR unhydrogenated, indicating 373 Ni/ZSM-5 specifically catalyzed H・・・H transfer to 9,10- 374 diphenylanthracene. In other words, 9,10-diphenylanthracene 375 hydrogenation can be used to justify the type of hydrogen 376
Tetrahydrofuran/Methanol Mixture−Insoluble Portion of
transfer, i.e., H• or H・・・H transfer.
377
 Pingshuo Bituminous Coal over an Iron−Sulfur System
Pingshuo Bituminous Coal over an Iron−Sulfur System
Ni/attapulgite powder is also active for arene hydrogenation 378
under mild conditions.78 Under 4 MPa of IHP for 4 h, 379 anthracene was completely converted to perhydroanthracenes at 380 175 °C, but anthracene conversion dropped to <75% with 9,10- 381 dihydroanthracene as the main product at 300 °C, clearly 382 indicating that saturated hydrogenation should not proceed at 383 high temperatures. Similar to other ultrafine metals, Ni/ 384 attapulgite powder also activates H2 to H・・・H to effectively 385 hydrogenate anthracene to perhydroanthracenes at low temper- 386 atures, but at high temperatures H・・・H bond tends to be 387 thermally cleaved to produce H•, which transfer to anthracene 388 predominantly yields 9,10-dihydroanthracene, and there is an 389 equilibrium between anthracene hydrogenation and 9,10- 390 dihydroanthracene dehydrogenation by the H• transfer, as 391
![]()
347 Different from metal sulfides, ultrafine metals mainly activates
•
392 s9
393
348 H2 to H・・・H under mild conditions.74,75 Di(1-naphthyl)
There are three problems for H
transfer to specifically cleave 394
349 methane was completely converted to its hydrogenated
350 products, which mainly consist of naphthylmethyltetralins and
351 ditetralylmethanes, under 10 MPa of IHP at 300 °C over
352 ultrafine iron, while adding sulfur inhibited di(1-naphthyl)
353 methane hydrogenation but promoted di(1-naphthyl) methane
354 hydrocracking.74 The main products from 9,10-diphenylan-
355 thracene hydrogenation are 9,10-diphenyl-1,2,3,4-tetrahydroan-
356 thracene and 9,10-diphenyl-1,2,3,4,5,6,7,8-octahydroanthra-
357 cene over ultrafine metals under mild conditions, since the
358 main active hydrogen is H・・・H and its transfer to 9- and 10-
359 positions is very difficult due to the much smaller distance
360 between the two hydrogen atoms in H・・・H than that between the
361 two carbon atoms in 9- and 10-positions of 9,10-diphenylan-
362 thracene.71,72 Ni/ZSM-5 prepared using nickel tetracarbonyl as
363 the precursor for nickel is highly active for completely 364 hydrogenating polymethylbenzenes,76 naphthalene, phenan- 365 threne, and anthracene under 6 MPa of IHP at 160−220 °C
366 and also effective for catalyzing coal tar hydrogenation.77 9,10-
367 Diphenylanthracene was hydrogenated to 9,10-diphenyl-
368 1,2,3,4,5,6,7,8-octahydroanthracene as the main product along
369 with 9,10-dicyclohexylanthracene, 9,10-dicyclohexyl-1,2,3,4-
370 tetrahydroanthracene, and 9-cyclohex- cyl-10-phenyl-
bridged linkages. The first one is the difficulty in significantly 395 increasing the H• concentration, because such an increase 396 enhances the possibility for scavenging the H• species by their 397
collision to produce H2. The second one results from the partial 398
hydrogenation by H• transfer, e.g., the CHC of di(1-naphthyl) 399
methane to naphthylmethyltetralins and bi(tetralyl) meth- 400 anes,54,74 1-benzylnaphthalene to benzyltetralins,53 and 1,2- 401 di(1-naphthyl) ethane to naphthylethyltetralins and 1,2- 402 (ditetralyl) ethanes.68 In particular, H• transfer to condensed 403 arenes usually produces multiple partially hydrogenated 404 products; e.g., H• transfer to 2,3-naphthacene over an activated 405 carbon produced seven partially hydrogenated products.62 406 Apparently, separating any partially hydrogenated product 407 from such a mixture is not easy. The last one is caused by the 408 scavenging effect of benzylic hydrogen on H• transfer, e.g., as 409 mentioned above, H• transfer over either a metal sulfide or an 410 activated carbon to the ipso-position of di(1-naphthyl) methane 411 induces di(1-naphthyl) methane hydrogenation.52−54,74,75 412 However, as Scheme 10 demonstrates,79 the abstraction of 413 s10 benzylic hydrogen from the − CH2− in di(1-naphthyl) methane 414
by H• and subsequent H• addition to the resulting −•CH− 415
should also be considered. Such an abstraction and addition 416
Scheme 10. Possible Mechanism for Scavenging H• by Benzylic Hydrogen in Di(1-naphthyl)methane79
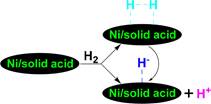
genation at 300 °C, suggesting that the new solid acid is active 448 for heterolytically splitting H2 to mobile H+ and immobile H− 449 attached on the surface of the new solid acid and H+ transfer to 450 the ipso-position of di(1-naphthyl) methane induces di(1- 451 naphthyl) methane hydrocracking. Interestingly, the yield of 452 naphthalene is appreciably higher than that of 1-methylnaph- 453 thalene, implying that the demethylation appreciably proceeded 454
 over the new solid acid.
over the new solid acid.
455
![]()
417 consumed H• without contribution to di(1-naphthyl) methane
418 hydrocracking.
419 Different from H• transfer, H+ transfer does not abstract
420 benzylic hydrogen and scavenging H+ does not occur by the
The CHC of an insoluble portion from Lingwu bituminous 456 coal was also investigated at 300 °C over the new solid acid.8 457 Compared to the NCHC, the CHC of the insoluble portion 458 produced much more arenes and phenols. Noteworthily, ca. 459 66.1% of the arenes is due to diphenylmethane, further 460 suggesting that some arenes can be released in a high yield 461 from the CHC of a heavy carbon resource similar to the CHC of 462
421 collision of H+ species to provide a possibility for significantly
the insoluble portion from Pingshuo bituminous coal.
Most of 463
422 increasing the H• concentration. For example, the acidities of
423 carborane acids80 and a mixture called magic acid81 consisting of
424 equimolar parts of FSO3H and SbF5 are 1 million and 1 billion
the resulting phenols are alkyl-substituted phenols. In the 464
insoluble portion from Lingwu bituminous coal, as Scheme 11 465 s11
exhibits, diphenylmethyl group (DPMG) could be connected 466
+
425 times, respectively, stronger than that of concentrated H2SO4. In
with a MMG-substituted anthracene ring (MMGSAR). H
467
426 addition, two or more H+ species cannot simultaneously attack
427 the same AR to avoid the AR hydrogenation.
428 Unfortunately, the practical application of both carborane
429 acids and magic acid in catalyzing the hydroconversion of heavy
430 carbon resources is difficult due to the extreme difficulty in
431 synthesizing carborane acids and too severe corrosiveness of
432 magic acid. Conveniently preparing a less corrosive, recyclable,
433 and highly active catalyst for heterolytically splitting H2 to
434 effectively release mobile H+ is one of our ultimate targets. We
435 believe that such a catalyst greatly facilitates the directional
436 CHC of heavy carbon resources under mild conditions.
437 A new solid acid was prepared by ultrasonically impregnating
438 isometric pentachloroantimony and trimethylsilyl trifluorome-
439 thanesulfonate into an activated carbon.82 Multiple analyses
440 reveal the strong interactions among pentachloroantimony,
441 trimethylsilyl trifluoromethanesulfonate, and the activated
442 carbon in the new solid acid and suggest that the new solid
transfer to the ipso-position of the MMGSAR induces the 468
cleavage of the DPMG-MMGSAR bond, releasing diphenylme- 469 thylium followed by the abstraction of H− by diphenylmethy- 470 lium from the surface of H−-attached new solid acid. Similarly, 471 the −CH2−O- bridged linkage connecting an alkyl-substituted 472 BR with a MMGSAR could be cleaved by H+ transfer to the 473 oxygen atom in the bridged linkage to release alkyl-substituted 474 phenols. Significantly higher yields of arenes and arenols were 475 also released from the CHC of insoluble portion obtaining from 476
Piliqing subbituminous coal over a highly active magnetic solid 477 superacid compared to the NCHC at 300 °C.83 More 478 interestingly, significantly high yield of mesitylene, a value- 479 added compound having many important applications, were 480
released from the CHC of insoluble portions obtaining from 481 Piliqing subbituminous coal,82 Hefeng subbituminous coal,84 482 Huozhou lignite, and Shaerhu lignite,85 and all the coal samples 483 were collected from Xinjiang Uyghur Autonomous Region, 484
443 acid exhibits an appreciably stronger acidity than either
China.
485
444 pentachloroantimony/activated carbon or trimethylsilyl tri-
445 fluoromethanesulfonate/activated carbon. Over the new solid
446 acid, di(1-naphthyl) methane was specifically hydrocracked to
447 naphthalene and 1-methylnaphthalene without NR hydro-
Naomaohu lignite is also a low-rank coal collected from 486 Xinjiang Uyghur Autonomous Region, China. Its CHC was 487 ultrasonically extracted with isometric carbon disulfide/acetone 488 mixed solvent (MS) and the resulting insoluble portion was 489
![]()
Scheme 11. Possible Pathways for Releasing Diphenylmethane and Alkyl-Substituted Phenols from the CHC of the Insoluble Portion Obtained from Lingwu Bituminous Coal over the New Solid Acid8
490 isolated into light and heavy insoluble portions in carbon
491 tetrachloride. The light insoluble portion was subjected to
492 NCHC and CHC in cyclohexane at 160 °C under 4 MPa of IHP
493 for 12 h. Trifluoromethanesulfonic acid supported on an acid-
494 treated attapulgite was used as the catalyst for the CHC. As a
495 result, the yield of soluble portion from the CHC is 60.0%, while
496 the yield (1.8%) of soluble portion from the NCHC is negligible.
497 The predominant products from the catalytic hydroconversion
ring. The >CH−OH2+ bond cleavage to produce H2O and 527 perhydroanthr-9-ylium is relatively easy because of the resulting 528 relatively stable perhydroanthr-9-ylium. Then, perhydroanthr-9- 529 ylium can abstract H− from the surface of H−-attached Ni/solid 530 acid to yield perhydroanthracene. By the synergic H+ and H・・・H 531 transfer, the CHCs of other heavy carbon resources, including 532 extracts from an oil sludge89 and Jinjitan subbituminous coal,91 533 and an organic waste oil91,92 to cyclanes over Ni/solid acids were 534
498 are oxygen-containing organic compounds, especially 4-
also achieved.
535
499 methylpent-3-en-2-one and 4-hydroxy-4-methyl-pentan-2-one,
500 suggesting that the cleavage of >Cα−Oβ− and >Cβ−Oα− bonds
501 in the light insoluble portion significantly proceeded during the
502 CHC.86 Oxygen-containing organic compounds are also
503 predominant from the CHC of an alkali lignin over Ni/Hβ.87
504 Nickel supported on solid acids proved to be the bifunctional
505 catalyst for the CHC of heavy carbon resources to cyclanes.88−92
506 Earlier investigation focused on the CHC of the methanol-
507 soluble portion, mainly consisting of heteroatom-containing
508 organic compounds with small amounts of chain alkanes,
509 alkenes, and arenes, from Xiaolongtan lignite in cyclohexane at
510 200 °C over Ni/Z5A.88 After the catalytic hydroconversion,
511 arenes and almost all the heteroatom-containing organic
512 compounds were converted to chain alkanes and cyclanes.
513 Especially, the resulting cyclanes account for 70% of the
514 products, while ca. 45% of the methanol-soluble portion is
515 arenols, which was completely converted to cyclanes.
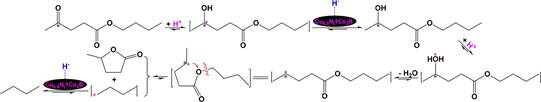
Chen et al.93 investigated CHC of butyl levulinate, an 536 important biomass-derived compound, to γ-valerolactone over 537 Cu0.5Ni1Co3B. Their results exhibited that the conversion of 538 butyl levulinate and γ-valerolactone reached up to 99.7% and 539 89.5%, respectively, under 3 MPa of H2 at 200 °C for 3 h, but 540 they did not explain the reaction pathway. As found by Scotti et 541 al.94 that very small particles (less than 5 nm) of metallic Cu 542 could exhibit catalytically relevant Lewis acidity. So, the 543 activation of H2 to H・・・H and heterolytic cleavage of H・・・H to 544
relatively mobile H+ and immobile H− attached on the 545
Cu0.5Ni1Co3B surface could also proceed. There are three 546 oxygen atoms in butyl levulinate and the conversion of butyl 547 levulinate to γ-valerolactone suggests that H+ preferentially 548 attacks the oxygen atom in acetyl group of butyl levulinate 549 followed by abstracting H− from the catalyst surface by the 550 protonated butyl levulinate to produce butyl 4-hydroxypenta- 551
+
s12 516 As illustrated in Scheme 12, Ni/solid acid simultaneously
noate, H
transfer to the oxygen atom in hydroxy group of butyl 552
![]() 517 activates H2 to H・・・H and splits
both H2 and H・・・H to mobile
H+
517 activates H2 to H・・・H and splits
both H2 and H・・・H to mobile
H+
4-hydroxypentanoate, dehydration from the protonated butyl 4- 553
hydroxypentanoate, and the formation of the >C*H−O− bond 554
Scheme 12. Catalysis of a Ni/Solid Acid in Activating H2 to
+
Furfural is another important biomass-derived compound and 556
H・・・H and Splitting both H2 and H・・・H to Mobile H
−
and
mainly obtained from corncob.95 It can be selectively hydro- 557
 Immobile H
Immobile H
Attached on the Surface of Ni/Solid Acid
genated to furan-2-ylmethanol, (tetrahydrofuran-2-yl) meth- 558 anol, 2-methylfuran, and cyclopentanone,96,97 and among them 559 furan-2-ylmethanol has multiple applications in producing 560 resins, biofuels, fuel additives, lysine, vitamin C, levulinic acid, 561 alkyl levulinates, and γ-valerolactone.98−100 Thereby, selective 562 catalytic hydrogenation of furfural to furan-2-ylmethanol 563 received great attention.95−122 In light of practical applications, 564 completely converting furfural to furan-2-ylmethanol is of great 565 importance to avoid the troublesome separation of target 566 product furan-2-ylmethanol from the reaction mixture, includ- 567
![]() 518 and immobile H− attached on
the surface of Ni/solid acid.
518 and immobile H− attached on
the surface of Ni/solid acid.
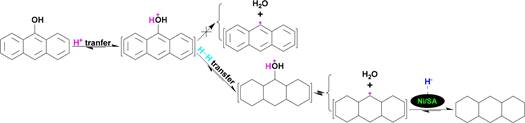
519 Taking anthracen-9-ol as an example, H+ transfer to the oxygen
520 atom in anthracen-9-ol cannot cleave the >Car−OH bond
521 because of the extreme labile leaving group, i.e., anthr-9-ylium
s13 522 (Scheme 13). However, the added H+ on the oxygen atom
523 avoids the Ni/solid acid poisoning by the oxygen atom and
524 ensures the saturated hydrogenation of the anthracene ring in
525 the protonated anthracen-9-ol to a protonated perhydroan-
526 thracen-9-ol via subsequent H・・・H transfer to the anthracene
ing the reactant furfural and its by products. Furfural 568 hydrogenation of furan-2-ylmethanol can significantly proceed 569 at near room temperature over rare earth element- or novel 570 metal-containing catalysts (entries 2, 6, 10, 14, and 17 in Table 571 t1 1), but the complete conversion of furfural to furan-2- 572 t1 ylmethanol was achieved only over Pt(3) Co(3)/C (Entry 6 573 in Table 1) in these cases and the high catalyst cost prevents the 574 catalysts from practical applications. The main solvents used 575 under optimal conditions include alkanols (entries 1, 4, 5, 7−9, 576
![]()
Scheme 13. Synergic H+ and H・・・H Transfer during the CHC of Anthracen-9-ol to Perhydroanthracene over a Ni/Solid Acid89
Scheme 14. Possible Pathway for the CHC of Butyl Levulinate to γ-Valerolactone
![]()
Table 1. Comparison of the Catalytic Hydrogenation of Furfural to Furan-2-ylmethanol under Different Conditionsa
![]()
catalyst solvent IHP (MPa) temperature (°C) time (h) FC (%) FMS (%) AH Co/SBA-15 ethanol 2 150 1.5 96 >95 NC Ru/UiO-66 water 0.5 20 4 94.9 100 NC Pd5%-Cu5%/MgO water 0.8 130 0.9 100 99 NC γ-Fe2O3@HAP IP 0 180 10 96.2 91.7 NC Cu/AC−SO3H IP 0.4 105 2 >99.9 >99.9 H+ Pt(3) Co(3)/C water 0.1 35 10 100 100 H+ Cu/MgAlO ethanol 4 150 3 >99 >99 NC Ni/NAC-1−1073 IP 4 80 3 100 ca. 100 NC CuCo0.4/C-873 ethanol 3 140 1 98.7 97.7 NC Co/ZrLa0.2Ox water 2 40 10 98 95 H+ Zr(OH)4 IP 0 170 2.5 100 98.9 H+ Ni3Fe1/SiO2 methanol 3.4 140 2 100 96.5 NC Ni@N/C-g-800 THFAS 2 140 6 99 98 NC Ir/H@MoOx-400 water 2 30 6 >99 >99 H+ Co@CPNs1.5−1.5−4 IP 3 180 2.5 99 99 NC Cu/MgO-Al2O3 IP 0 210 1 100 89.3 H+ Pt(3) Ni(3)/C water 2 35 12 99 93 NC Cu/C IP 2 175 2 99.8 100 H+ HT_MgFe-3 IP 0 170 6 ca. 97 ca. 90 H− Cu−Co/C IP 3 200 2.5 100 97.2 H+ Co−N−C-700 FA 0 150 6 100 99.9 H+ Ni0.09Zn/NC600 IP 2 170 2 99.7 100 H+ Co17Zn/NC600 ethanol 2 125 2.5 100 100 H+
1100
2101
3102
4103
5104
6105
7106
8107
9108
10109
11110
12111
13112
14113
15114
16115
17116
18117
19118
20119
21120
22121
23122
aFC, FMS, AH, NC, IP, THFAS, and FA denote furfural conversion, furan-2-ylmethanol selectivity, active hydrogen, not clarified, 2-propanol, tetrahydrofuran aqueous solution, and formic acid, respectively.
![]()
577 11, 12, 15, 16, 18−20, 22, and 23 in Table 1), formic acid (entry
578 21 in Table 1), tetrahydrofuran aqueous solution (entry 13 in
579 Table 1), and water (entries 2, 3, 6, 10, 14, and 17 in Table 1).
600
Superbases were extensively used for catalyzing organic 601
580 The heterolytic cleavage of H2 and the transfer of the resulting
581 H+ to the oxygen atom in the −CHO group of furfural were
and CO2 capture,
but only a few attempts 602
582 clarified to be key steps for furfural hydrogenation to furan-2-
583 ylmethanol (entries 5, 6, 10, 14, and 18−23 in Table 1) under
584 pressurized H2. Without H2, the transfer of H+ from a solvent
585 also induced furfural hydrogenation to furan-2-ylmethanol over
586 an acidic catalyst (entries 4, 11, 16, 19, and 21 in Table 1).
587 Taking 2-propanol as an example, similar to H+ transfer from
588 2-propanol to vanillin displayed in Scheme 5, the strong
589 adsorption of the oxygen atom in the hydroxy group of 2-
590 propanol on the surface of an acidic catalyst leads to the
591 heterolytic cleavage of −O−H bond to release H+ and retain the
592 isoproxy anion on the catalyst surface. The H− transfer to the
have been tried on the application of superbases in the CHC of 603 LRMCs129−134 and heavy carbon resources.130−135 Since the 604 C−O bridged bonds (COBBs), such as ARCH2O−CH2AR, 605 AROCH2AR, and ARO-AR, are the predominant COBBs in 606
lignites,136−138 insight into the catalytic hydroconversions of 607
LRMCs, such as oxybis(methylene) dibenzene, benzyloxyben- 608 zene, and oxidibenzene with COBBs, has become a powerful 609 tool to understand the mechanisms for selectively cleaving 610 COBBs and hydrogenating AR in lignites at molecular level.10,12 611 Among the COBBs, BRO-BR bond (360 kJ mol−1) in 612 oxidibenzene is the strongest,139,140 so the research gaining 613 insight into the cleavage of COBB in oxidibenzene has attracted 614
593 oxygen atom in the hydroxy group of furfural produces
extensive attention.
615
594 furyl(hydroxy) methylium, followed by H− abstraction from
595 the tertiary carbon in the isoproxy anion by furyl(hydroxy)
596 methylium to generate furan-2-ylmethanol. Co17Zn/NC600
597 proved to be an effective non-novel catalyst for completely
598 hydrogenating furfural to furan-2-ylmethanol (entry 23 in Table
599 1).
Our earlier investigation used oxidibenzene, benzyloxyben- 616 zene, and oxybis(methylene) dibenzene as lignin-related model 617 compounds (LRMCs) and compared the activities and 618 selectivities of Ni/γ-Al2O3, Mg2Si/γ-Al2O3, and Ni−Mg2Si/γ- 619
Al2O3 for the CHC of the LRMCs.129 As a result, Ni−Mg2Si/γ- 620
Al2O3 proved to be the most effective for converting 621
622 oxidibenzene to cyclohexane, benzyloxybenzene to cyclohexane 623 and methylcyclohexane, and oxybis(methylene) dibenzene to 624 methylcyclohexane at 240 °C by the synergic transfer of H− and 625 H・・・H to the LRMCs. The CHC of oxidibenzene over Mg2Si/γ- 626 Al2O3 only produced benzene and phenol, and phenol 627 conversion to benzene also proceeded without H・・・H trans- 628 fer,129,130 while oxidibenzene was not converted at all over
As the main compounds in the MS-soluble portion (MSSP) 649 from Piliqing subbituminous coal, nonsubstituted arenes were 650 effectively converted to nonsubstituted cyclanes133 and a series 651 of normal alkanes were also released56 during the CHC at 240 652
°C over Ni−Mg2Si/γ-Al2O3, while substituted phenols and 653 benzenediols are the main products from the CHC of 654 Xiaolongtan lignite at 300 °C over Fe O @SiO @mSiO .134 655
3 4 2 2
629 trifluoromethanesulfonic acid/attapulgite powder due to the
The much weaker alkalinity of Fe3O4@SiO2@mSiO2 than 656
630 resulting extremely labile phenylium from H+ transfer to the
Mg Si/γ-Al O
and the lack of metallic nickel in Fe O @SiO @ 657
2 2 3
3 4 2
s15 631 oxygen atom in oxidibenzene (Scheme 15).130 Although the
mSiO2
lead to the difficulty in deoxygenation and subsequent 658
![]() BR hydrogenation of the resulting substituted phenols and 659
BR hydrogenation of the resulting substituted phenols and 659
Scheme 15. Difference between H− Transfer and H+ Transfer
benzenediols.
660
 in Cleaving
the >Car−O− Bond in Oxydibenzene
in Cleaving
the >Car−O− Bond in Oxydibenzene
Wang et al.142 investigated the catalytic hydrogenolysis of a 661 poplar wood sawdust-derived lignin over a NiAl alloy. Their 662 results showed that 18.9% and 55.5% of the lignin were 663 converted to monomers and oligomers, respectively, in 0.2 mol 664 L−1 of NaOH aqueous solution under 2 MPa of IHP at 220 °C 665 for 3 h. They tried to reveal the mechanism for the catalytic 666 hydrogenolysis by examining the depolymerization of 2- 667
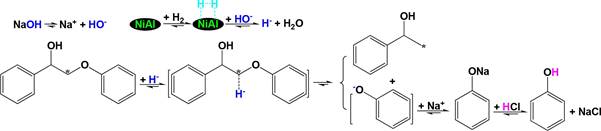
phenoxy-1-phenylethanol over the NiAl alloy in NaOH aqueous 668 solution under pressurized H2 at 110 °C for 1 h. As a result, 669 phenol and 1-phenylethan-1-ol were detected as the main 670 products. In the reaction systems of both catalytic hydro- 671 genolysis of the lignin and 2-phenoxy-1-phenylethanol depoly- 672 merization, in addition to H・・・H resulting from H2 activation by 673
the NiAl alloy, the main active hydrogen should be H− rather 674
![]() than H+, due to the use of NaOH aqueous solution as the 675 solvent. As Scheme 16 illustrates, the reaction of HO− in the 676 s16 NaOH aqueous solution
with H・・・H produces H− and H2O, and 677 the resulting H− tends to attack the *C, which is positively 678 charged,
to cleave the *C−O- bond and produce 1-phenylethan- 679
than H+, due to the use of NaOH aqueous solution as the 675 solvent. As Scheme 16 illustrates, the reaction of HO− in the 676 s16 NaOH aqueous solution
with H・・・H produces H− and H2O, and 677 the resulting H− tends to attack the *C, which is positively 678 charged,
to cleave the *C−O- bond and produce 1-phenylethan- 679
1-ol and phenoxy anion, followed by the reaction of the phenoxy 680
+
632 >Car−O− bond is much stronger than the −CH2−O− bond in
anion with Na
to produce sodium phenolate and the 681
633 benzyloxybenzene, H− transfer only induces the cleavage of 634 >Car−O− bond in benzyloxybenzene because the Car in >Car− 635 O− is most positively charged141 and much more tends to be 636 attacked by H−131 under mild conditions.
acidification of sodium phenolate with HCl aqueous solution 682
to produce phenol and NaCl. The main problem of such a 683 process for future practical application is the consumption of 684 NaOH and HCl aqueous solution along with the production of 685
637 The NCHC and CHC of MS-insoluble portion (MSISP)
NaCl-containing wastewater.
686
638 from Naomaohu lignite were compared at 240 °C under 4 MPa 639 of IHP.56 The yield of soluble portion from the CHC over Ni− 640 Mg2Si/γ-Al2O3 is much higher than that from the NCHC and 641 the main products in the soluble portion from the CHC are 642 chain alkanes and cyclanes, while the NCHC mainly produced 643 arenes and oxygen-containing organic compounds. In contrast, 644 the CHC of MSISP from Naomohu lignite mainly produced 645 oxygen-containing organic compounds over a Ni-free magnetic 646 solid superbase,132 further illustrating that the synergic transfer 647 of H− and H・・・H is crucial for deoxygenation under mild 648 conditions.
Aqueous coprecipitation followed reduction with H2 is an 687
important approach for preparing highly active supported 688 metallic catalysts, in which ultrafine metal particles are highly 689 dispersed onto the support surface.143−146 Using this method, 690
NiCa/Al2O3 was prepared and used as a catalyst for the CHC of 691
MSISPs from Runbei lignite and Shaerhu subtuminous coal at 692
180 °C.147 The yields of MSSPs from Runbei lignite and 693 Shaerhu subtuminous coal are 16.5% and 11.8%, respectively, 694 while those from the CHC under 1 MPa of IHP are 31.1% and 695 27.4%, respectively, i.e., the total yields of soluble portions by the 696
extraction and CHC are 47.6% and 39.2% based on the organic 697
![]()
Scheme 16. Possible Pathways for the Formation of H・・・H and H− and Subsequent H− Transfer to *C in 2-Phenoxy-1- phenylethanol to Induce 2-Phenoxy-1-phenylethanol Depolymerization
698 matters in Runbei lignite and Shaerhu subtuminous coal, 699 respectively. Interestingly, only alkanes and arenes were 700 detected in the MSSPs from the CHC and their compositions 701 (in total 16 normal alkanes listed in Table S1, 5 branched alkanes
resources, (2) lack of deep insight into the features of 736
macromolecular structures of the heavy carbon resources, and 737
(3) less attention paid to the mechanisms for the formation of 738
different active hydrogen species and their different roles in 739
702 listed in Table S2, and 47 arenes listed in Table S3) are
directionally converting heavy carbon resources.
740
703 significantly simpler than those of coal tar and coal liquefaction
704 oil, not only enabling insight into the macromolecular structures 705 of the MSISPs but also facilitating the separation of the products. 706 ARs, including BR, NR, phenanthrene ring, anthracene ring, 707 pyrene ring, BR-BR, and BR-NR, in the precursors of the alkanes 708 and arenes could be connected with bridged linkages of
Although the macromolecular compositions of different heavy 741 carbon resources could be quite different, they have some similar 742 unit structures, i.e., ARs connected by bridged linkages and with 743 side chains on most of the ARs. Heteroatoms are contained in 744 some ARs, bridged linkages, and side chains, and numerous 745 soluble organic species are embedded into the complex 746
709 −(CH2)m−O−(CH2)n− and/or side chains of CH3(CH2)m−
macromolecular networks, as illustrated in Figure 3.149
747 f3
710 O−(CH2)n−. In NiCa/Al2O3, Ni activates H2 to H・・・H and Ca
711 splits H・・・H to mobile H− and immobile H+. H− transfer to the
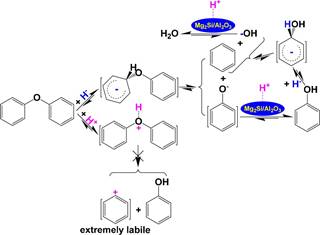
712 oxygen atom in AR(CH2)m−O−(CH2)nAR and CH3(CH2)m− 713 O−(CH2)nAR and subsequent H+ abstraction from the surface 714 of H+-attached NiCa/Al2O3 release AR(CH2)m‑1CH3, AR- 715 (CH2)n‑1CH3, and CH3(CH2)mCH3. However, at 180 °C 716 under 4 MPa of IHP, only alkanes and alkyl-substituted cyclanes 717 were detected in the products from the NiCa/Al2O3-catalyzed 718 hydroconversion of the MSISPs, indicating that H・・・H transfer 719 to the ARs in the resulting alkyl-substituted moieties 720 predominantly proceeded.
721 Further investigation for preparing extremely active and 722 recyclable solid acids will facilitate cleaving most of the alkyl 723 groups on the ARs by effective H+ transfer to the ipso-positions 724 of the ARs to obtain nonsubstituted arenes, especially
MS proved to be a powerful solvent for effectively extracting 748 the soluble organic species from the macromolecular networks 749 under ultrasonic irradiation.89,150,151 As a result, the organic 750 matter in heavy carbon resources can be isolated into soluble 751 portion and insoluble portion. A number of organic compounds 752
were significantly enriched and even separated as pure 753 compounds by subsequent fine separations, including fractional 754 extraction, gradient column chromatography, and sequential 755
crystallization from the soluble portion.152−159 Alternatively, the 756
soluble portion can be converted to alkanes as well as 757 nonsubstituted and alkyl-substituted cyclanes under pressurized 758 H2 over Ni-loading catalysts.57,58,88,135,160 The insoluble portion 759 can be ultrasonically isolated to a light insoluble portion and a 760 heavy insoluble portion in carbon tetrachloride according to the 761
725 nonsubstituted CAs, with significantly simpler compositions
density difference.
The catalytically hydroconverted light 762
726 for subsequent separation to obtain value-added pure arenes.
727 Developing smart technologies for the CHC of heavy carbon 728 resources to value-added products, especially condensed 729 aromatics and cyclanes,5,81,148 under mild conditions is of 730 great importance for efficiently utilizing heavy carbon resources 731 to create great social wealth with near-zero emission of emission
732 of exhaust gas, wastewater, and waste residue. The key 733 challenges facing the development include: (1) the high 734 contents of ARs and heteroatoms in addition to the insoluble 735 and complex macromolecular compositions of the heavy carbon
insoluble portion can be relatively easily separated from the 763
catalyst by the density difference, and adding a magnetic core 764 into the catalyst further facilitates recovering the cata- 765 lyst.83,119,131−135,161 In addition, ultrasonically extracting a 766
solid sample, e.g., a coal sample, significantly reduced the size 767 of the sample particles,150 facilitating the CHC. Using an 768 insoluble portion as the reactant makes an insight into the 769 mechanisms for the CHC easier by avoiding the disturbance of 770
the inherently existing soluble species in the raw sample. 771 Furthermore, removing soluble species from the raw sample also 772 makes the separation of a reaction mixture from the CHC easier. 773 Distinguishing the type of active hydrogen species is very 774 important for understanding the hydrogen transfer mechanism 775
![]()
![]()
![]()
![]() 776 to provide scientific basis for
constructing a highly active
777 catalyst. Benzylic hydrogen, i.e., α-H, is easily identified 778
according the sample structure. As mentioned above, catalytic 779 hydrogenation
of 9,10-diphenylanthracene under mild con-
780 ditions provides
a proper evidence
for H・・・H or H• transfer, since 781 H• transfer predominantly
proceeds to 9- and 10-positions in 782 9,10-diphenylanthracene,
while H・・・H transfer preferentially
783 occurs to other positions.71,72,77 According to the products
from 784
the catalytic hydrocracking of benzyloxybenzene under mild 785 conditions, especially below
200 °C at which benzyloxybenzene 786
is inert toward
thermolysis or by H• transfer, H+ transfer, and H− 787 transfer can be clearly
distinguished. In detail, H+ transfer 788 predominantly
proceeds to the oxygen atom in benzyloxyben- 789 zene
to break the −O−CH2−
bond and yield phenol and
776 to provide scientific basis for
constructing a highly active
777 catalyst. Benzylic hydrogen, i.e., α-H, is easily identified 778
according the sample structure. As mentioned above, catalytic 779 hydrogenation
of 9,10-diphenylanthracene under mild con-
780 ditions provides
a proper evidence
for H・・・H or H• transfer, since 781 H• transfer predominantly
proceeds to 9- and 10-positions in 782 9,10-diphenylanthracene,
while H・・・H transfer preferentially
783 occurs to other positions.71,72,77 According to the products
from 784
the catalytic hydrocracking of benzyloxybenzene under mild 785 conditions, especially below
200 °C at which benzyloxybenzene 786
is inert toward
thermolysis or by H• transfer, H+ transfer, and H− 787 transfer can be clearly
distinguished. In detail, H+ transfer 788 predominantly
proceeds to the oxygen atom in benzyloxyben- 789 zene
to break the −O−CH2−
bond and yield phenol and
790 toluene as the initial products162 because the oxygen atom is
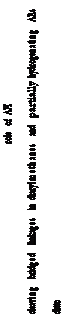 791 most negatively charged141 and
the resulting tolylium is 792 relatively stable, while H− transfer selectively occurs
to the 793 ipso-carbon in the oxygen atom-connected BR of benzylox- 794 ybenzene to cleave the >Car−O−
bond and generate benzene
795 and phenylmethanol as the initial products,130 due to
the most 796 positive
charged ipso-carbon.141
791 most negatively charged141 and
the resulting tolylium is 792 relatively stable, while H− transfer selectively occurs
to the 793 ipso-carbon in the oxygen atom-connected BR of benzylox- 794 ybenzene to cleave the >Car−O−
bond and generate benzene
795 and phenylmethanol as the initial products,130 due to
the most 796 positive
charged ipso-carbon.141
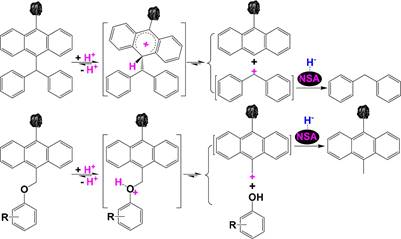
t2 797 As summarized in Table 2, both metal sulfides and carbon 798 materials can catalyze the formation of H•, but their catalyzes are 799 different. Over a metal sulfide, e.g., FeS2, H2 is converted to H• 800 via a chain reaction shown in Scheme 5, while H• results from
801 the homolytic splitting of H2 over a carbon material. H2 can be 802 activated to H・・・H over an ultrafine metal at temperatures lower 803 than 200 °C, but the resulting H・・・H can be easily split to H• at 804 temperatures higher than 300 °C. H• transfer to an ipso-position 805 in a diarylmethane induces the cleavage of AR-CH2AR bond due
806 to the formation of the relatively stable.CH2AR, while H•
807 transfer to a condensed AR only leads to the partial
![]()
![]()
![]()
![]() 808 hydrogenation
of the condensed AR, e.g., the partial
hydro- 809
genation of anthracene to 9,10-dihydroanthracene rather than 810 the
saturated hydrogenation of anthracene to perhydroanthra-
811 cenes due to the low H•-accepting ability of the residual BRs in 812 9,10-dihydroanthracene
and the stronger tendence for H• to 813
abstract α-H from
9,10-dihydroanthracene.78 Solid acids and 814 solid bases tend to heterolytically split H2 to release
H+ and H−, 815 respectively, and both H+
and H− transfer
can induce the
808 hydrogenation
of the condensed AR, e.g., the partial
hydro- 809
genation of anthracene to 9,10-dihydroanthracene rather than 810 the
saturated hydrogenation of anthracene to perhydroanthra-
811 cenes due to the low H•-accepting ability of the residual BRs in 812 9,10-dihydroanthracene
and the stronger tendence for H• to 813
abstract α-H from
9,10-dihydroanthracene.78 Solid acids and 814 solid bases tend to heterolytically split H2 to release
H+ and H−, 815 respectively, and both H+
and H− transfer
can induce the
![]()
![]()
![]() 816 cleavage of bridged linkages
between ARs and side chain on the 817 ARs, and
the removal of some heteroatoms, but the positions
818 accepting H+ are
quite different from those accepting H+ to
816 cleavage of bridged linkages
between ARs and side chain on the 817 ARs, and
the removal of some heteroatoms, but the positions
818 accepting H+ are
quite different from those accepting H+ to
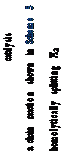
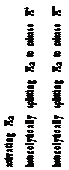 s17 819 produce different products (Scheme 17). Both Ni/solid
acids 820 and
Ni/solid bases have bifunctional catalyzes, i.e., Ni/solid 821 acids activates H2 to H・・・H and heterolytically splits H2 and H・・・ 822 H to release
H+, while Ni/solid bases
activates H2 to H・・・H and 823 heterolytically splits H2 and H・・・H to release H−. The synergic
s17 819 produce different products (Scheme 17). Both Ni/solid
acids 820 and
Ni/solid bases have bifunctional catalyzes, i.e., Ni/solid 821 acids activates H2 to H・・・H and heterolytically splits H2 and H・・・ 822 H to release
H+, while Ni/solid bases
activates H2 to H・・・H and 823 heterolytically splits H2 and H・・・H to release H−. The synergic
824 transfer of either H・・・H and H+ or H・・・H and H− leads to the
![]() 825 effective cleavage of some
bridged linkages between ARs and 826 some side chains on ARs, the saturated hydrogenation, and 827 heteroatom removal.
825 effective cleavage of some
bridged linkages between ARs and 826 some side chains on ARs, the saturated hydrogenation, and 827 heteroatom removal.
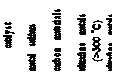
![]()
![]()
![]() 828 Elaborately constructing highly active solid acids, solid bases, 829 Ni/solid acids, and Ni/solid bases is of great importance. Over 830 the catalysts, many bridged linkages and even
some side chains 831 can be cleaved,
and many heteroatoms outside and on ARs can 832 be removed by the CHC of light
insoluble portions from heavy
833 carbon resources
under mild conditions to get soluble
portions 834 with relatively simple
composition, facilitating subsequent
835 separation to obtain pure chemicals, especially value-added 836 chemicals,
such as condensed aromatics. The remaining 837
economically inseparable species can be converted to value- 838 added
cyclanes by subsequent catalytic hydrofining.
828 Elaborately constructing highly active solid acids, solid bases, 829 Ni/solid acids, and Ni/solid bases is of great importance. Over 830 the catalysts, many bridged linkages and even
some side chains 831 can be cleaved,
and many heteroatoms outside and on ARs can 832 be removed by the CHC of light
insoluble portions from heavy
833 carbon resources
under mild conditions to get soluble
portions 834 with relatively simple
composition, facilitating subsequent
835 separation to obtain pure chemicals, especially value-added 836 chemicals,
such as condensed aromatics. The remaining 837
economically inseparable species can be converted to value- 838 added
cyclanes by subsequent catalytic hydrofining.
839 Directionally converting heavy carbon resources to more soluble 840 portion with simpler composition under mild conditions is a 841 crucial step for efficiently using heavy carbon resources.
Feng-Yun Ma − Laboratory of Coal Clean Conversion & Chemical Engineering Process, College of Chemistry and Chemical Engineering, Xinjiang University, Urumqi 830046 Xinjiang Uyghur Autonomous Region, China
873
874
875
876
842 Compared to H• transfer, the synergies between H+ and H・・・
843 H transfer and between H− and H・・・H transfer are significantly
Zhi-Min Zong − Jiangsu Province Engineering Research Center 877
of Fine Utilization of Carbon Resources and Key Laboratory of 878
844 more effective for the process. Enhancing the synergies depends
Coal Processing and Efficient Utilization, Ministry of
879
845 on the construction of extremely active and recyclable catalysts,
846 over which H2 can be effectively split and/or activated to
Education, China University of Mining & Technology, Xuzhou 880
![]() 221116 Jiangsu, China; orcid.org/0000-0002-3144-4558 881
221116 Jiangsu, China; orcid.org/0000-0002-3144-4558 881
847 produce high concentrations of H+ and H・・・H or H− and H・・・H
848 to directly crop the macromolecules in heavy carbon resources.
Wei Zhao − Jiangsu Province Engineering Research Center of
![]() Fine Utilization of Carbon
Resources and Key Laboratory of Coal Processing and Efficient Utilization, Ministry of
Fine Utilization of Carbon
Resources and Key Laboratory of Coal Processing and Efficient Utilization, Ministry of
882
883
884
|
850 *sı Supporting Information
851 The Supporting Information is available free of charge at
Education, China University of Mining & Technology, Xuzhou 885 221116 Jiangsu, China; orcid.org/0000-0002-5559-8957 886 Zhong-Hai Ni − Jiangsu Province Engineering Research Center 887 of Fine Utilization of Carbon Resources and Key Laboratory of 888
852 https://pubs.acs.org/doi/10.1021/acs.energyfuels.3c01713.
Coal Processing and Efficient Utilization, Ministry of
889
853 Tables of alkanes and arenes detected in SPsCHC of RL
854 and SSBC (PDF)
Education, China University of Mining & Technology, Xuzhou 890
![]() 221116 Jiangsu, China; orcid.org/0000-0002-8706-2583 891
221116 Jiangsu, China; orcid.org/0000-0002-8706-2583 891
|
856 Corresponding Author
857 Xian-Yong Wei − Key Laboratory of Chemistry and Chemical
Xing Fan − Key Laboratory of Chemistry and Chemical
![]() Engineering on Heavy-Carbon Resources, Yili Normal University,
Yining 835000 Xinjiang, China; orcid.org/ 0000-0001-5530-5563
Engineering on Heavy-Carbon Resources, Yili Normal University,
Yining 835000 Xinjiang, China; orcid.org/ 0000-0001-5530-5563
Lin-Bing Sun − State Key Laboratory of Materials-Oriented
892
893
894
895
896
858 Engineering on Heavy-Carbon Resources, Yili Normal
Chemical Engineering, Jiangsu National Synergetic Innovation 897
859 University, Yining 835000 Xinjiang, China; Laboratory of 860 Coal Clean Conversion & Chemical Engineering Process, 861 College of Chemistry and Chemical Engineering, Xinjiang
Center for Advanced Materials, College of Chemical
![]() Engineering, Nanjing Tech
University, Nanjing 211816, China; orcid.org/0000-0002-6395-312X
Engineering, Nanjing Tech
University, Nanjing 211816, China; orcid.org/0000-0002-6395-312X
898
899
900
862 University, Urumqi 830046 Xinjiang Uyghur Autonomous
Jing-Pei Cao − Jiangsu Province Engineering Research Center of 901
863 Region, China; Jiangsu Province Engineering Research Center
864 of Fine Utilization of Carbon Resources and Key Laboratory of
Fine Utilization of Carbon Resources and Key Laboratory of
Coal Processing and Efficient Utilization, Ministry of
902
903
865 Coal Processing and Efficient Utilization, Ministry of
866 Education, China University of Mining & Technology, Xuzhou
Education, China University of Mining & Technology, Xuzhou 904
![]() 221116 Jiangsu, China; orcid.org/0000-0002-1544-7441 905
221116 Jiangsu, China; orcid.org/0000-0002-1544-7441 905
![]() 867 221116
Jiangsu, China; orcid.org/0000-0001-7106-
867 221116
Jiangsu, China; orcid.org/0000-0001-7106-
868 4624; Email: wei_xianyong@163.com
Yun-Peng Zhao − Laboratory of Coal Clean Conversion &
Chemical Engineering Process, College of Chemistry and Chemical Engineering, Xinjiang University, Urumqi 830046
906
907
908
869 Authors
Xinjiang Uyghur Autonomous Region, China; Jiangsu Province 909
870 Xiang Bai − Key Laboratory of Chemistry and Chemical
Engineering Research Center of Fine Utilization of Carbon
910
871 Engineering on Heavy-Carbon Resources, Yili Normal
872 University, Yining 835000 Xinjiang, China
Resources and Key Laboratory of Coal Processing and Efficient 911
Utilization, Ministry of Education, China University of Mining 912
|
913 |
& Technology, Xuzhou 221116 Jiangsu, China; |
orcid.org/ Xiang Bai received his Bachelor’s degree of science from Jingchu 974 |
||
|
914 |
University of Technology in 2013 and a Master’s degree of engineering 975 |
|||
|
915 Shi-Chao Qi − State Key Laboratory of Materials-Oriented from Xinjiang University in 2016. He currently works at Yili Normal 976 |
||||
|
916 |
Chemical Engineering, Jiangsu National Synergetic Innovation |
University and is studying for a Doctoral degree of science at Xinjiang |
977 |
|
|
917 |
Center for Advanced Materials, College of Chemical |
University with the major research interest of chemistry and chemical |
978 |
|
|
918 |
Engineering, Nanjing Tech University, Nanjing 211816, |
engineering of heavy carbon resources. |
979 |
|
![]()
![]() 919 China;
orcid.org/0000-0002-9609-7710
919 China;
orcid.org/0000-0002-9609-7710
920 Jing Liang − Jiangsu Province Engineering Research Center of 921 Fine Utilization of Carbon Resources and Key Laboratory of 922 Coal Processing and Efficient Utilization, Ministry of
923 Education, China University of Mining & Technology, Xuzhou
924 221116 Jiangsu, China
925 Xiao-Ming Yue − Jiangsu Province Engineering Research Center
926 of Fine Utilization of Carbon Resources and Key Laboratory of
Feng-Yun Ma received her Bachelor’s degree of Engineering from East 980 China University of Science and Technology in 1982 and Master’s 981 degree of engineering from Dalian University of Technology in 2002. 982 She worked at Former Xinjiang Institute of Technology (now Xinjiang 983 University) as a Teaching Assistant during 1982-1987, Lecturer during 984 1988−1994, Associate Professor during 1994−1999, and Professor 985 during 1999−2020 with the major research interest of coal clean 986
927 Coal Processing and Efficient Utilization, Ministry of
conversion and chemical engineering process.
987
928 Education, China University of Mining & Technology, Xuzhou
929 221116 Jiangsu, China
930 Fang-Jing Liu − Laboratory of Coal Clean Conversion &
931 Chemical Engineering Process, College of Chemistry and
932 Chemical Engineering, Xinjiang University, Urumqi 830046 933 Xinjiang Uyghur Autonomous Region, China; Jiangsu Province 934 Engineering Research Center of Fine Utilization of Carbon
Zhi-Min Zong received her Bachelor’s degree of science from Xuzhou 988 Normal College in 1982, a Master’s degree of engineering from The 989 University of Tokyo in 1992, and Doctoral degree of engineering from 990 China University of Mining & Technology in 1997. She worked at 991 China University of Mining & Technology as a Lecturer during 1993− 992 1998, Associate Professor during 1998−2002, and Professor during 993
2002−2020 with the major research interest of directional hydro- 994
935 Resources and Key Laboratory of Coal Processing and Efficient
936 Utilization, Ministry of Education, China University of Mining
conversion of heavy carbon resources.
995
![]() 937 & Technology,
Xuzhou 221116 Jiangsu, China; orcid.org/
937 & Technology,
Xuzhou 221116 Jiangsu, China; orcid.org/
![]() 939 Wen-Long Mo − Laboratory of Coal Clean Conversion & 940 Chemical Engineering Process, College of Chemistry and 941 Chemical
Engineering, Xinjiang University, Urumqi 830046 942 Xinjiang
Uyghur Autonomous Region, China; orcid.org/
939 Wen-Long Mo − Laboratory of Coal Clean Conversion & 940 Chemical Engineering Process, College of Chemistry and 941 Chemical
Engineering, Xinjiang University, Urumqi 830046 942 Xinjiang
Uyghur Autonomous Region, China; orcid.org/
Wei Zhao received her Bachelor’s degree of science from Nanjing 996
University in 1988 and Master’s and Doctoral degrees of engineering 997 from China University of Mining & Technology (CUMT) in 2003 and 998 2009, respectively. She worked at CUMT as a Lecturer during 2000− 999 2003, Associate Professor during 2003−2011, and Professor from 2011 1000 with the major research interest of catalytic conversion of heavy carbon 1001
944 Jing-Mei Liu − Laboratory of Coal Clean Conversion &
945 Chemical Engineering Process, College of Chemistry and 946 Chemical Engineering, Xinjiang University, Urumqi 830046 947 Xinjiang Uyghur Autonomous Region, China
948 Yu-Hong Kang − School of Chemistry and Chemical
949 Engineering, Yulin University, Yulin 719000 Shanxi, China
950 Guang-Hui Liu − School of Chemistry and Chemical
951 Engineering, Yulin University, Yulin 719000 Shanxi, China;
Zhong-Hai Ni received his Bachelor’s and Master’s degrees of 1003 engineering from China University of Mining and Technology 1004 (CUMT) in 2000 and 2003, respectively, and a Doctoral degree of 1005 science from Tsinghua University in 2006. He worked at Shandong 1006 University as a Lecturer during 2006-2008 and an Associate Professor 1007 in 2009 and at CUMT as a Professor from 2010 with the major research 1008 interest of chemistry and chemical engineering of coal tar-enriched 1009
953 Zhong-Qiu Liu − Key Laboratory of Life-Organic Analysis of
954 Shandong Province, School of Chemistry and Chemical 955 Engineering, Qufu Normal University, Qufu 273165 956 Shandong, China
957 Li Li − Jiangsu Province Engineering Research Center of Fine
958 Utilization of Carbon Resources and Key Laboratory of Coal
Xing Fan received his Bachelor’s degree of science and Master’s degree 1011 of engineering from University of Science and Technology of China in 1012 2000 and 2004, respectively, and Doctoral degree of philosophy from 1013 Louisiana State University in 2010. He worked at Shandong University 1014 of Science and Technology as a Professor since 2019 with the major 1015 research interest of chemistry of heavy carbon resources. He is an 1016
959 Processing and Efficient Utilization, Ministry of Education,
960 China University of Mining & Technology, Xuzhou 221116
Emeritus Professor of Yili Normal University.
1017
961 Jiangsu, China
962 Complete contact information is available at:
963 https://pubs.acs.org/10.1021/acs.energyfuels.3c01713
964 Notes
965 The authors declare no competing financial interest.
Lin-Bing Sun received his Bachelor’s and Master’s degrees of 1018
engineering from China University of Mining & Technology in 2002 1019 and 2005, respectively. He received his Ph.D. from Nanjing University 1020 in 2008. Then, he joined Nanjing Tech University and is a Professor of 1021 the university with current research interests focusing on fabrication of 1022 porous functional materials and their applications in adsorption and 1023 catalysis. He was a postdoctoral research associate at Texas A&M 1024
966 Biographies
University in 2011−2012.
1025
967 Xian-Yong Wei received his Bachelor’s degree of science from Nanjing 968 University in 1982 and his Master’s and Doctoral degrees of 969 engineering from The University of Tokyo in 1989 and 1992, 970 respectively. He worked at China University of Mining & Technology 971 as a professor from 1993 to 2023 with the major research interest of 972 chemistry and chemical engineering of heavy carbon resources. He
Jing-Pei Cao received his Bachelor’s degree of science and Master’s 1026 degree of engineering from China University of Mining and 1027 Technology (CUMT) in 2004 and 2007, respectively, and a Doctoral 1028 degree of engineering from Gunma University in 2011. He worked at 1029 CUMT as an Associate Professor during 2012−2015 and a Professor 1030 since 2016. His research interest is value-added utilization of coals and 1031
973 currently works at Yili Normal University as an emeritus professor.
biomass.
1032
1033 Yun-Peng Zhao received his Bachelor’s degree of engineering from
from 2023 with the major research interest of efficient utilization of 1093
1034 Zhengzhou University in 2005 and Doctoral degree of engineering from
heavy carbon resources.
1094
1035 Dalian University of Technology in 2010. He worked at China 1036 University of Mining & Technology as a Lecturer during 2011−2014 1037 and Associate Professor during 2014−2021, and has been working at 1038 the university as a Professor from 2021. His major research interests are 1039 thermal conversion of coals and biomass.
1040 Shi-Chao Qi received his Bachelor’s degree of science from China
Zhong-Qiu Liu received his Bachelor’s degree of science from Jining 1095 University in 2011, Master’s degree of engineering from Jiangsu Normal 1096 University in 2014, and Doctoral degree of engineering from China 1097 University of Mining & Technology in 2018. He worked at Qufu 1098 Normal University as a Lecturer during 2018−2022 and an Associate 1099 Professor since 2022 with the major research interest of directional 1100
1041 University of Petroleum in 2011, Master’s degree of engineering from
hydroconversion of heavy carbon resources.
1101
1042 China University of Mining & Technology in 2014, and Doctoral 1043 degree of engineering from Kyushu University in 2017. Then he joined 1044 Nanjing Tech University as a lecturer has been working at the university
Li Li received her bachelor of Science in 2010, Master’s degree of 1102
engineering in 2013, and Doctoral degree of engineering in 2023 from 1103
China University of Mining & Technology. Her major research focuses 1104
1045 as an Associate Professor from 2020. His current research area is
1046 advanced porous materials, heterogeneous catalysis, quantum chemical
1047 calculation, and molecular simulation.
1048 Jing Liang received her Bachelor’s and Master’s degrees of science from
on directional hydroconversion of heavy carbon resources.
1105
1106
1049 Soochow University in 1996 and 1999, respectively, and Doctoral 1050 degree of engineering from China University of Mining & Technology 1051 (CUMT) in 2015. She worked at Xuzhou Medical University as a 1052 lecturer during 2000−2004, at CUMT as a Lecturer during 2004−2008 1053 and an Associate Professor from 2009. Her current research area is 1054 green organic synthesis and catalytic conversion of heavy carbon 1055 resources.
Our related work was subsidized by the National Basic Research 1107
Program of China (Grants 2004CB217801, 2004CB217601, 1108 2007CB210205, and 2011CB201302), the National Natural 1109 Science Foundation of China (Grants 29676045, 20076051, 1110
90410018, 20676142, 20776149, 20936007, 50921002, 1111
51221462, 51074153, 51134021, 21206187, 21206188, 1112
U1503293, 21676292, 21776298, and U1710102), and the 1113
National Natural Science Foundation of Yili Normal University 1114
1056 Xiao-Ming Yue received her Bachelor’s and Doctoral degrees of
1057 engineering from China University of Mining & Technology (CUMT) 1058 in 2007 and 2012, respectively. She worked at CUMT as a Lecturer 1059 during 2012−2016 and an Associate Professor from 2017 with current
(Grant 22XKZY07).
1115
1116
1060 interests focusing on advanced porous materials and catalytic
1061 conversion of coals.
1062 Fang-Jing Liu received his Bachelor’s degree of science and Master’s 1063 and Doctoral degrees of engineering from China University of Mining 1064 & Technology (CUMT) in 2009, 2012, and 2016, respectively. Then 1065 he worked at University of Wyoming as a postdoc associate scientist. He
1066 joined CUMT as a Lecturer and worked at CUMT as an Associate
ARs, aromatic rings; BR, benzene ring; CHC, catalytic 1117
hydroconversion; COBBs, C−O bridged bonds; DPMG, 1118 diphenylmethyl group; IHP, initial hydrogen pressure; MS, 1119 isometric carbon disulfide/acetone mixed solvent; MSISP, MS- 1120 insoluble portion; MSSP, MS-soluble portion; LRMCs, lignin- 1121 related model compounds; MMG, macromolecular group; 1122 MMGSAR, MMG-substituted anthracene ring; NCHC, non- 1123
1067 Professor from 2020. His current research area is coal structure and coal
1068 directional conversion to valued-added chemicals.
1069 Wen-Long Mo received his bachelor and doctoral degrees of
catalytic hydroconversion; NR, naphthalene ring
1124
1125
1070 engineering from Xinjiang University in 2011 and 2016, respectively.
(1) Wei, X. Y.; Zong, Z. M.; Sun, L. B.; Qin, Z. H.; Zhao, W. Scientific 1126
foundation for efficient utilization of heavy carbon resources. Chem. Ind. 1127
1071 He worked at Xinjiang University as a Lecturer during 2016−2017 and
Eng. Pro. 2006, 25, 1134−1142.
1128
1072 Associate Professor from 2018 with the major research interest of
1073 heterogeneous catalysis, advanced porous materials, and chemical
(2) Wei, X. Y. Technological innovation for efficient utilization of 1129
heavy carbon resources, 2020 Int. Forum Petrochem. Green Dev. October 1130
1074 engineering of heavy carbon resources.
18 & 19, 2020, Xuzhou, China.
1131
1075 Jing-Mei Liu received bachelor, master, and doctoral degrees of
1076 engineering from Xinjiang University in 2006, 2009, and 2019,
(3) Wei, X. Y. Technology development for efficient utilization of 1132
heavy carbon resources, 2020 Int. Forum Petrochem. Green Dev. April 17, 1133
2020, Shanghai, China.
1134
1077 respectively. She worked at Xinjiang University as a assistant
1078 experimentalist during 2009-2013, experimentalist during 2014-2019, 1079 and senior experimentalist since 2020 with the major research interest 1080 of clean conversion of coals.
(4) Wei, X. Y.; Zong, Z. M.; Zhao, W.; Ni, Z. H.; Cao, J. P.; Fan, X.; 1135 Zhao, Y. P.; Zhao, X. Y.; Liang, J.; Liu, F. J.; Kang, Y. H.; Ma, F. Y.; 1136 Aisha, N.; Mo, W. L.; Liu, J. M. Chemical technology development for 1137 efficient utilization of heavy carbon resources, The 2nd Int. Conf. Appl. 1138
1081 Yu-Hong Kang received his Bachelor’s degree of engineering from
Chem. Ind. Catal., October 16−19, 2020, Dalian, China.
1139
1082 Henan University in 2007, Master’s degree of engineering from
(5) Wei, X. Y.; Zong, Z. M.; Fan, X.; Li, Z. K. Chapter 17. Coal-based 1140
products and their uses. In: Coal Production and Processing Technology. 1141
1083 Taiyuan University of Technology in 2012, and Doctoral degree of
CRC Press: 2015; pp 383−412.
1142
1084 engineering from China University of Mining & Technology in 2021.
1085 He worked at Yulin University as a Teaching Assistant during 2012−
1086 2014, Lecturer during 2014−2018, and Associate Professor since 2018
(6) Sun, L. B.; Zong, Z. M.; Kou, J. H.; Cao, J. P.; Yu, G. Y.; Zhao, W.; 1143 Li, B. M.; Lee, C. W.; Xie, K. C.; Wei, X. Y. Identification of organic 1144 chlorines and iodines in the extracts from hydrotreated Argonne 1145
1087 with the major research interest of chemical engineering of heavy
Premium coal residues. Energy Fuels 2007, 21, 2238−2239.
1146
1088 carbon resources.
1089 Guang-Hui Liu received his Bachelor’s degree of engineering from
(7) Sun, L. B.; Wei, X. Y.; Liu, X. Q.; Zong, Z. M.; Li, W. Release of 1147
organonitrogen and organosulfur compounds during hydrotreatment 1148
of Pocahontas No. 3 coal residue over an activated carbon. Energy Fuels 1149
1090 Tongji University in 2014 and doctoral degree of engineering from
2009, 23, 5284−5286.
1150
1091 China University of Mining & Technology in 2020. Then he joined
1092 Yulin University, where he has been working as an Associate Professor
(8) Xiao-Ming, Y.; Xian-Yong, W.; Bing, S.; Ying-Hua, W.; Zhi-Min, 1151
Z.; Zi-Wu, L. Solid superacid-catalyzed hydroconversion of an 1152
1153 extraction residue from Lingwu bituminous coal. Int. J. Mining Sci.
(29)
Yang, X.; Shao, S.; Li, X.; Tang, D. Catalytic transfer 1220
1154 Technol. 2012, 22, 251−254.
hydrogenation of bio-oil over biochar-based CuO catalyst using 1221
1155
(9) Yu, L. C.; Wei, X. Y.; Wang, Y. H.; Zhang, D. D.; Wen, Z.; Zong, Z.
methanol as hydrogen donor. Renew. Energy 2023, 211, 21−30.
1222
1156 M.; Fan, X.; Zhao, Y. P.; Zhao, W.; Zhu, Y. L. Catalytic hydroconversion 1157 of extraction residue from Shengli lignite over Fe-S/ZSM-5. Fuel 1158 Process. Technol. 2014, 126, 131−137.
(30) Ai, L.; Shi, H.; Chen, J. In-situ deoxygenation of methyl palmitate 1223
to hydrocarbons on highly dispersed Ni−Re/TiO2 catalyst in aqueous 1224
phase using methanol as a hydrogen donor. J. Energy Inst. 2023, 109, 1225
1159
(10) Kuhlmann, E. J.; Jung, D. Y.; Guptill, R. P.; Dyke, C. A.; Zang, H.
101265.
1226
1160 K. Coal liquefaction using a hydrogenated creosote oil solvent: H-atom 1161 transfer from hydrogen donor components in the solvent. Fuel 1985, 1162 64, 1552−1557.
(31) Lu, H. Y.; Wei, X. Y.; Yu, R.; Peng, Y. L.; Qi, X. Z.; Qie, L. M.; 1227
Wei, Q.; Lv, J.; Zong, Z. M.; Zhao, W.; Zhao, Y. P.; Ni, Z. H.; Wu, L. 1228
Sequential thermal dissolution of Huolinguole lignite in methanol and 1229
1163
(11) Bate, K.; Harrison, G. Fate of hydrogen-donor molecules in two-
in ethanol. Energy Fuels 2011, 25, 2741−2745.
1230
1164 stage liquefaction using model solvents. Fuel 1992, 71, 289−293.
(32) Chen, B.; Wei, X. Y.; Zong, Z. M.; Yang, Z. S.; Qing, Y.; Liu, C. 1231
1165
(12) Sudakova, I. G. Influence of hydrogen donor solvent on kinetic
Difference in chemical composition of the products from supercritical 1232
1166 features of hydrogenation of some fluid products of coal liquefaction.
methanolysis of two lignites. Appl. Energy 2011, 88, 4570−4576.
1233
1167 Coal Sci. Technol. 1995, 24, 1491−1494.
(33) Chen, B.; Wei, X. Y.; Yang, Z. S.; Liu, C.; Fan, X.; Qing, Y.; Zong, 1234
1168
(13) Liu, J.; Zhao, L.; Liu, Z.; Liu, Q. Catalyst-free liquefaction of
Z. M. ReaxFF reactive force field for molecular dynamics simulations of 1235
1169 lignin for monophenols in hydrogen donor solvents. Fuel Process.
lignite depolymerization in supercritical methanol with lignite-related 1236
1171
(14) Shimada, I.; Uno, C.; Watanabe, Y.; Takatsuka, T. Catalytic
(34) Li, Z. K.; Zong, Z. M.; Yan, H. L.; Wang, Y. G.; Wei, X. Y.; Shi, D. 1238
1172 cracking of three-ring polycyclic aromatic hydrocarbons in the presence
1173 of hydrogen donors. Fuel Process. Technol. 2022, 232, 107267.
L.; Zhao, Y. P.; Zhao, C. L.; Yang, Z. S.; Fan, X. Alkanolysis simulation 1239
of lignite-related model compounds using density functional theory. 1240
1174
(15) Liu, M.; Yang, J.; Li, Y.; Liu, Z.; Liu, Q.; Shi, L. Radical reactions
Fuel 2014, 120, 158−162.
1241
1175 and two-step kinetics of sub-bituminous coal liquefaction in various
1176 solvents. Energy Fuels 2019, 33 (3), 2090−2098.
(35) Li, Z. K.; Zong, Z. M.; Yan, H. L.; Wang, Y. G.; Ni, H. X.; Wei, X. 1242
Y.; Li, Y. H. Characterization of acidic species in ethanol-soluble 1243
1177
(16) Sheng, Q.; Wang, G.; Zhang, Q.; Gao, C.; Ren, A.; Duan, M.;
portion from Zhaotong lignite ethanolysis by negative-ion electrospray 1244
1178 Gao, J. Seven-lump kinetic model for non-catalytic hydrogenation of
ionization Fourier transform ion cyclotron resonance mass spectrom- 1245
1179 asphaltene. Energy Fuels 2017, 31 (5), 5037−5045.
etry. Fuel Process. Technol. 2014, 128, 297−302.
1246
1180
(17) Yung, M. M.; Starace, A. K.; Mukarakate, C.; Crow, A. M.;
(36) Yang, H. M.; Zhao, W.; Norinaga, K.; Fang, J. J.; Wang, Y. G.; 1247
1181 Leshnov, M. A.; Magrini, K. A. Biomass catalytic pyrolysis on Ni/ZSM-
1182 5: Effects of nickel pretreatment and loading. Energy Fuels 2016, 30 (7),
Zong, Z. M.; Wei, X. Y. Separation of phenols and ketones from bio-oil 1248
produced from ethanolysis of wheat stalk. Sep. Purif. Technol. 2015, 152, 1249
1183 5259−5268.
238−245.
1250
1184
(18) Zong, Z. M.; Zhang, W. H.; Jiang, Q.; Lu, J.; Wei, X. Y.
(37) Liu, F. J.; Wei, X. Y.; Zong, Z. M.; Fan, M. H. Characterization of 1251
1185 Photochemical reactions of hydroarenes with N-bromosuccinimide.
the oxygenated chemicals produced from supercritical methanolysis of 1252
1186 Bull. Chem. Soc. Jpn. 2002, 75, 769−771.
modified lignites. Energy Fuels 2016, 30, 2636−2645.
1253
1187
(19) McMillen, D. F.; Malhotra, R.; Nigenda, S. E. The case for
(38) Yan, H. L.; Zong, Z. M.; Li, Z. K.; Kong, J.; Zheng, Q. X.; Zhao, 1254
1188 induced bond scission during coal pyrolysis. Fuel 1989, 68, 380−386.
M. X.; Li, Y.; Wei, X. Y. Insight into the chemical complexity of soluble 1255
1189
(20) Zong, Z. M.; Wei, X. Y. Effects of molecular hydrogen and
portions from cornstalk methanolysis. Energy Fuels 2016, 30, 3020− 1256
1191 Fuel Process. Technol. 1994, 41, 79−85.
(39) Hu, L.; Guo, X. H.; Wei, X. Y.; Liu, F. J.; Xu, M. L.; Liu, T. L.; 1258
1192
(21) Wei, X. Y.; Ogata, E.; Zong, Z. M.; Niki, E. Inhibiting effect of
Zhang, F. B. Research on the influence of sequential isopropanolysis 1259
1193 hydrogen-donating compounds on1,3-diphenylpropane. Coal Conv.
liquefaction on the composition of liquid tars and physicochemical 1260
1195
(22) McMillen, D. F.; Malhotra, R.; Chang, S. J.; Ogier, W. C.;
(40) Bai, Y.; Cui, K.; Sang, Y.; Wu, K.; Yan, F.; Mai, F.; Ma, Z.; Wen, 1262
1196 Nigenda, S. C.; Fleming, R. H. Mechanisms of hydrogen transfer and
1197 bond scission of strongly bonded coal structures in donor-solvent
Z.; Chen, H.; Chen, M.; Li, Y. Catalytic Depolymerization of a lignin- 1263
rich corncob residue into aromatics in supercritical ethanol over an 1264
alumina-supported NiMo alloy catalyst. Energy Fuels 2019, 33 (9), 1265
1199
(23) Futamura, S.; Koyanagi, S.; Kamiya, Y. The hydrogenolysis of
(41) Li, C.; Zhang, M.; Li, H.; Zhang, Y. Renewable tar-derived Pd@ 1267
1200 diarylmethane. Mechanism for hydrogen transfer from solvents and
biocarbon for mild and efficient selectively hydrodeoxygenation of 1268
1201 additive effects of hydrogen shuttlers and organic radical sources. Fuel
vanillin. Energy Fuels 2021, 35 (5), 4169−4181.
1269
1203
(24) Hu, J.; Zhao, M.; Jiang, B.; Wu, S.; Lu, P. Catalytic transfer
transfer process to coal and coal extract. Ind. Eng. Chem. Process. Des. 1271
1205 bimetallic catalyst. Energy Fuels 2020, 34 (8), 9754−9762.
(43) Kamiya, Y.; Nagae, S. Relative reactivity of hydrogen donor 1273
1206
(25) Song, L.; Ouyang, Y.; Huang, S.; Li, Z.; Sun, M. Insight into
solvent in coal liquefaction. Fuel 1985, 64, 1242−1245.
1274
1207 liquefaction process of sawdust with hydrogen donor solvents. Biomass
(44) Chiba, K.; Tagaya, H.; Saito, N. Liquefaction of Yallourn coal by 1275
1209
(26) Zhang, J.; Li, C.; Hu, S.; Gu, J.; Yuan, H.; Chen, Y. Mechanistic
(45)
Woodfine, B.; Steedman, W.; Kemp, W. Donor solvent 1277
1210 insights into copper oxides catalyzed bio-based furfural hydrogenation
interactions during coal liquefaction. Fuel 1989, 68, 293−297.
1278
1211 using methanol as in-situ hydrogen donor. Renew. Energy 2022, 200,
(46) Bate, K.; Harrison, G. Fate of hydrogen-donor molecules in two- 1279
1213
(27)
Mu, D.; Li, Z.; Yu, S.; Liu, S. Hydrodechlorination of
(47) Park, H. B.; Kim, K. D.; Lee, Y. K. Promoting asphaltene 1281
1214 chlorophenols with methanol as hydrogen donor over carbon nanotube
conversion by tetralin for hydrocracking of petroleum pitch. Fuel 2018, 1282
1216
(28)
Philippov, A. A.; Anufrieva, V. M.; Pakharukova, V. P.;
(48) Miura, K.; Shimada, M.; Mae, K.; Sock, H. Y. Extraction of coal 1284
1217 Martyanov, O. N. Donor activity of sub- and supercritical primary
1218 alcohols in catalyst-free and Ni/γ-Al2O3 catalyzed transfer hydro-
below 350 °C in flowing non-polar solvent. Fuel 2001, 80, 1573−1582. 1285
(49) Ouchi, K.; Makabe, M. Hydrogen transfer in the hydrogenation 1286
1219 genation of furfural. J. Supercrit. Fluid 2023, 193, 105815.
of model compounds. Fuel 1988, 67, 1536−1541.
1287
(50) Niu, B.; Jin, L. J.; Li, Y.; Shi, Z. W.; Li, Y. T.; Hu, H. Q.
(70) Herndon, W. C. Resonance energies of hydrocarbon radicals. 1356
1289 Mechanism of hydrogen transfer and role of solvent during heating-up
radical reactivities of polycyclic aromatic hydrocarbons. J. Org. Chem. 1357
1290 stage of direct coal liquefaction. Fuel Process. Technol. 2017, 160, 130−
1981, 46, 2119−2125.
1358
1291 135.
(71) Zhou, S. L.; Ni, Z. H.; Yuan, X. H.; Zong, Z. M.; Wei, X. Y. 1359
1292
(51) Wei, X. Y.; Ogata, E.; Niki, E. FeS2-catalyzed hydrocracking of
Catalyses of metals and their sulfides in selective hydrogenation of 9,10- 1360
1293 di(1-naphthyl)methane. Bull. Chem. Soc. Jpn. 1992, 65, 987−990.
diphenylanthracene. J. China Univ. Mining Technol. 2001, 11, 80−82.
1361
1294
(52) Wei, X. Y.; Ogata, E.; Niki, E. FeS2-catalyzed hydrocracking of
(72) Zhou, S. L.; Ni, Z. H.; Yuan, X. H.; Zong, Z. M.; Wei, X. Y. Study 1362
1295 diarylmethanes. Sekiyu Gakkaishi 1992, 35, 358−361.
on the mechanism of catalytic hydrogenation of 9,10-diphenylan- 1363
1296
(53) Wei, X. Y.; Zong, Z. M. Solvent effect on diphenylmethane
thracene by GC-MS. J. Fuel Chem. Technol. 2001, 29, 80−82.
1364
1297 hydrocracking. Energy Fuels 1992, 6, 236−237.
(73) Wei, X. Y.; Zong, Z. M.; Zhao, W.; Cao, J. P.; Liu, Z. W.; Fan, X.; 1365
1298
(54) Wei, X. Y.; Ogata, E.; Zong, Z. M.; Niki, E. Effects of iron catalyst
Zhao, Y. P.; Zhao, X. Y. Directional conversion of coals: challenges and 1366
1299 precursors, sulfur, hydrogen pressure and solvent type on the
prospects. 2017 Int. Conf. Coal Sci. Technol. & 2017 Australia-China 1367
1300 hydrocracking of di(1-naphthyl)methane. Fuel 1993, 72, 1547−1552.
Symp. Energy, September 25−29, 2017, Beijing, China, No. O5−4,.
1368
1301
(55) Li, Z. K.; Wei, X. Y.; Yan, H. L.; Yu, X. Y.; Zong, Z. M.
(74) Wei, X. Y.; Ogata, E.; Niki, E. Catalyses of Fe and FeS2 on the 1369
1302 Characterization of soluble portions from thermal dissolution of
1303 Zhaotong lignite in cyclohexane and methanol. Fuel Process. Technol.
1304 2016, 151, 131−138.
reaction of di(1-naphthyl)-methane. Chem. Lett. 1991, 20, 2199−2202. 1370
(75) Wei, X. Y.; Ni, Z. H.; Zong, Z. M.; Zhou, S. L.; Xiong, Y. C.; 1371
Wang, X. H. Reaction of di(1-naphthyl)methane over metals and metal- 1372
1305
(56) Li, X. K.; Zong, Z. M.; Chen, Y. F.; Yang, Z.; Liu, G. H.; Liu, F. J.;
sulfur systems. Energy Fuels 2003, 17, 652−657.
1373
1306 Wei, X. Y.; Wang, B. J.; Ma, F. Y.; Liu, J. M. Catalytic hydroconversion
1307 of Yinggemajianfeng lignite over difunctional Ni-Mg2Si/γ-Al2O3. Fuel
(76) Qi, S. C.; Wei, X. Y.; Zong, Z. M.; Hayashi, J. I.; Yuan, X. H.; Sun, 1374
L. B. A highly active Ni/ZSM-5 catalyst for complete hydrogenation of 1375
1309
(57) Kang, Y. H.; Wei, X. Y.; Liu, G. H.; Ma, X. R.; Gao, Y.; Li, X.; Li, Y.
(77) Qi, S. C.; Wei, X. Y.; Hayashi, J. I.; Zong, Z. M.; Guo, L. L.; et al. 1377
1310 J.; Ma, Y. J.; Yan, L.; Zong, Z. M. Catalytic hydroconversion of ethanol-
Deep hydrogenation of coal tar over Ni/ZSM-5 catalyst. RSC Adv. 1378
1312 extraction residue to clean liquid fuel over a Y/ZSM-5 composite
1313 zeolite-supported nickel catalyst. Energy Fuels 2020, 34, 4799−4807.
(78) Liu, Z. Q.; Wei, X. Y.; Liu, F. J.; Wang, B. J.; Zong, Z. M. 1380
Temperature-controlled hydrogenation of anthracene over nickel 1381
1314
(58) Kang, Y. H.; Wei, X. Y.; Liu, G. H.; Gao, Y.; Li, Y. J.; Ma, X. R.;
nanoparticles supported on attapulgite powder. Fuel 2018, 223, 222− 1382
1315 Zhang, Z. F.; Zong, Z. M. Catalytic hydroconversion of soluble portion
1316 in the extraction from Hecaogou subbituminous coal to clean liquid fuel
229.
(79)
1383
Wei, X. Y. Chemical basis for the directional catalytic 1384
1317 over a Y/ZSM-5 composite zeolite-supported nickel catalyst. Fuel 2020,
hydroconversion of heavy carbon resources. The First Forum on Green 1385
1318 269, 117326.
Chemical Technology Development; Shanghai, China, 2019.
1386
1319
(59) Kang, Y. H.; Ma, Z. Y.; Zhang, X. Q.; Wei, X. Y.; Li, Y. J.; Liu, G.
(80) Reed, C. A.; Kim, K. C.; Bolskar, R. D.; Mueller, L. J. Taming 1387
1320 H.; Wang, A. M.; Ma, X. R.; Yan, L.; Zong, Z. M.; Bai, H. C.
superacids: stabilization of the fullerene cations HC60 and C60 . Science 1388
1322 coal and its residues by multiple technical strategies. Fuel 2022, 309,
1323 122111.
(81) Wei, X Y. Catalytic hydroconversion of low-rank coals under 1390
mild conditions and the synthesis of CO2-based polymers. The 358th 1391
1324
(60) Li, L.; Wei, X. Y.; Shao, C. W.; Yin, F.; Sun, B. K.; Liu, F. J.; Li, J.
symposium on Green and Low Carbon Development of Coal Engineering 1392
1325 H.; Liu, Z. Q.; Zong, Z. M. Honeycomb-like N/O self-doped
Science and Technology, Chinese Acadymy of Engineering & Summit of the 1393
1326 hierarchical porous carbons derived from low-rank coal and its
6th Coal Science and Technology; Beijing, China, 2023.
1394
1327 derivatives for high-performance supercapacitor. Fuel 2023, 331,
1328 125658.
(82) Yue, X. M.; Wei, X. Y.; Sun, B.; Wang, Y. H.; Zong, Z. M.; Fan, X.; 1395
Liu, Z. W. A new solid acid for specifically cleaving the Car-Calk bond in 1396
1329
(61) Ni, Z. H.; Zong, Z. M.; Zhang, L. F.; Sun, L. B.; Liu, Y.; Yuan, X.
di(1-naphthyl)methane. Appl. Catal. A Gen. 2012, 425−426, 79−84.
1397
1330 H.; Wei, X. Y. Synergic effect of sulfur on activated carbon-catalyzed
1331 hydrocracking of di(1-naphthyl)methane. Energy Fuels 2003, 17, 60−
1332 61.
(83) Yang, Z.; Wei, X. Y.; Zhang, M.; Teng, D. G.; Wang, Y. N.; Liu, Z. 1398
Q.; Zong, Z. M. Enhanced hydrocracking Car-Calk bridged bonds in the 1399
extraction residue from Piliqing subbituminous coal over a recyclable 1400
1333
(62) Sun, L. B.; Zong, Z. M.; Kou, J. H.; Zhang, L. F.; Ni, Z. H.; Yu, G.
and active magnetic solid superacid. Fuel Process. Technol. 2018, 176, 1401
1335 hydrogenation of polycylic arenes. Energy Fuels 2004, 18, 1500−1504.
(84) Yang, Z.; Wei, X. Y.; Li, Z. X.; Zhang, M.; Zong, Z. M.; Wei, Z. H.; 1403
1336
(63) Sun, L. B.; Zong, Z. M.; Kou, J. H.; Liu, G. F.; Sun, X.; Wei, X. Y.;
Li, Y. MOFs-derived N-doped carbon matrix superacid-catalyzed 1404
1337 Zhou, G. J.; Lee, C. W. Activated carbon-catalyzed hydrogen transfer to
hydrocracking of a residue from thermal dissolution of Hefeng 1405
1338 α,ω-diarylalkanes. Energy Fuels 2005, 19, 1−6.
subbituminous coal. Fuel Process. Technol. 2018, 180, 180−188.
1406
1339
(64) Sun, L. B.; Wei, X. Y.; Liu, X. Q.; Zong, Z. M.; Li, W.; Kou, J. H.
(85) Zhao, M. X.; Yang, N.; Xie, H. S.; Zhao, Z.; Zong, Z. M.; Wei, X. 1407
1340 Selective hydrogen transfer to anthracene and its derivatives over an
Y. Catalytic hydroconversion of extraction residues from two Chinese 1408
1341 activated carbon. Energy Fuels 2009, 23, 4877−4882.
coals to arenes. Fuel 2021, 284, 118910.
1409
1342
(65)
Farcasiu, M.; Smith, C. Modeling coal liquefaction. 1.
(86) Wu, Z. F.; Wei, X. Y.; Mo, W. L.; Kang, Y. H.; Zhang, X. Q.; Shan, 1410
1343 decomposition of 4-(1-naphthylmethyl)bibenzyl catalyzed by carbon
1344 black. Energy Fuels 1991, 5, 83−87.
X. K.; Liu, G. H.; Fan, X. Catalytic hydroconversion of the light residue 1411
from Yinggemajianfeng lignite over a solid superacid. Fuel 2022, 310, 1412
1345
(66) Farcasiu, M.; Petrosius, S. C.; Eldredge, P. A.; Anderson, R. R.;
122470.
1413
1346 Ladner, E. P. Modeling coal liquefaction. 3. catalytic reactions of
1347 polyfunctional compounds. Energy Fuels 1994, 8, 920−924.
(87) Zhou, X.; Wei, X. Y.; Ma, Y. M.; Zong, Z. M. Highly selective 1414
catalytic hydrocracking C-O bridged bonds in an alkali lignin over Ni/ 1415
1348
(67) Farcasiu, M.; Kaufman, P. B.; Ladner, E. P.; Derbyshire, F.;
Hβ. Fuel 2021, 287, 119474.
1416
1349 Jagtoyen, M. Carbon catalysts for reactions relevant to coal liquefaction.
1350 Proc. Int. Conf. Coal Sci. 1995, 24, 1303−1306.
(88) Li, W. T.; Wei, X. Y.; Liu, X. X.; Guo, L. L.; Qi, S. C.; Li, Z. K.; 1417
Zhang, D. D.; Zong, Z. M. Catalytic hydroconversion of methanol- 1418
1351
(68) Wei, X. Y.; Ogata, E.; Niki, E. FeS2-catalyzed hydrocracking of
soluble portion from Xiaolongtan lignite over difunctional Ni/Z5A. 1419
1352 α,ω-diarylalkanes. Bull. Chem. Soc. Jpn. 1992, 65, 1114−1119.
Fuel Process. Technol. 2016, 148, 146−154.
1420
1353
(69) Yonezawa, T.; Nagata, C.; Kato, H.; Imamura, A.; Morokuma, K.
(89) Kang, Y. H.; Wei, X. Y.; Li, J.; Jin, H. T.; Li, T.; Lu, C. Y.; Ma, X. 1421
1354 Guide to Quantum Chemistry; Kagaku-Dojin Press: Kyoto, Japan, 1990;
1355 pp 203−233.
R.; Zong, Z. M. Green and effective catalytic hydroconversion of an 1422
extractable portion from an oil sludge to clean jet and diesel fuels over a 1423
1424 mesoporous Y zeolite-supported nickel catalyst. Fuel 2021, 287,
1425 119396.
(107) Gong, W. B.; Chen, C.; Zhang, H. M.; Zhang, Y.; Zhang, Y. X.; 1492
Wang, G. Z.; Zhao, H. J. Selective liquid phase hydrogenation of furfural 1493
1426
(90) Gao, J.; Wei, X. Y.; Kang, Y. H.; Zhang, X. Q.; Wang, A. M.; Li, X.
to furfuryl alcohol by Ru/Zr-MOFs. J. Mol. Catal. A-Chem. 2017, 429, 1494
1427 L.; Gao, Y.; Liu, G. H.; Chen, T.; Li, Y. J.; Ma, X. R.; Bai, J. J.; Bai, N.;
51−59.
1495
1428 Ma, Y. J.; Zong, Z. M. Mesoporous zeolite Beta supported Ni 1429 nanoparticles for the catalytic hydroconversion of extraction portion 1430 from Jinjitan subbituminous coal to cycloalkanes. Fuel 2023, 339,
(108) Wang, Y.; Miao, Y. N.; Li, S.; Gao, L. J.; Xiao, G. M. Metal- 1496 organic frameworks derived bimetallic Cu-Co catalyst for efficient and 1497 selective hydrogenation of biomass-derived furfural to furfuryl alcohol. 1498
1431 127435.
Mol. Catal. 2017, 436, 128−137.
1499
1432
(91) Li, L.; Wei, X. Y.; Shao, C. W.; Li, Z.; Li, J. H.; Fan, Z. C.; Lu, K.
(109) Ma, Y. F.; Xu, G. Y.; Wang, H.; Wang, Y. X.; Zhang, Y.; Fu, Y. 1500
1433 L.; Liu, F. J.; Kong, Q. Q.; Zhao, J.; Liu, Z. Q.; Zong, Z. M. Catalytic
1434 hydroconversion of an organic waster oil to cyclanes over a supported
Cobalt nanocluster supported on zrrenox for the selective hydro- 1501
genation of biomass derived aromatic aldehydes and ketones in water. 1502
1436
(92) Li, L.; Wei, X. Y.; Liu, G. H.; Li, Z.; Li, J. H.; Liu, F. J.; Kong, Q.
(110) Zhang, J.; Dong, K. J.; Luo, W. N.; Guan, H. F. Selective transfer 1504
1437 Q.; Fan, Z. C.; Zong, Z. M.; Bai, H. C. Selective catalytic
1438 hydroconversion of organic waster oil to cyclanes over a coal fly ash-
hydrogenation of furfural into furfuryl alcohol on Zr-containing 1505
catalysts using lower alcohols as hydrogen donors. ACS Omega 2018, 1506
1441
(93) Chen, B.; Guo, H.; Wan, Z.; Xu, X.; Zhang, H.; Zhao, D.; Chen,
selective NiFe/SiO2 bimetallic catalyst with optimized solvent effect for 1509
1442 X.; Zhang, N. Efficient catalytic hydrogenation of butyl levulinate to γ-
the liquid-phase hydrogenation of furfural to furfuryl alcohol. ACS 1510
1443 valerolactone over a stable and magnetic CuNiCoB amorphous alloy
Sustainable Chem. Eng. 2018, 6 (10), 13287−13295.
1511
1444 catalyst. Energy Fuels 2018, 32 (4), 5527−5535.
(112) Nandi, S.; Saha, A.; Patel, P.; Khan, N. H.; Kureshy, R. I.; Panda, 1512
1445
(94) Scotti, N.; Dangate, M.; Gervasini, A.; Evangelisti, C.; Ravasio,
A. B. Hydrogenation of furfural with nickel nanoparticles stabilized on 1513
1446 N.; Zaccheria, F. Unraveling the role of low coordination sites in a Cu
1447 metal nanoparticle: A step toward the selective synthesis of second
nitrogen-rich carbon core−shell and its transformations for the 1514
synthesis of γ-valerolactone in aqueous conditions. ACS Appl. Mater. 1515
1448 generation biofuels. ACS Catal. 2014, 4 (8), 2818−2826.
Interfaces 2018, 10 (29), 24480−24490.
1516
1449
(95) Mariscal, R.; Maireles-Torres, P.; Ojeda, M.; Sádaba, I.; López-
(113) Xie, L. F.; Chen, T.; Chan, H. C.; Shu, Y. J.; Gao, Q. S. 1517
1450 Granados, M. Furfural: a renewable and versatile platform molecule for
1451 the synthesis of chemicals and fuels. Energy Environ. Sci. 2016, 9 (4),
Hydrogen doping into MoO3 supports toward modulated metal- 1518
support interactions and efficient furfural hydrogenation on iridium 1519
1453
(96) Lange, J. P.; van der Heide, E.; van Buijtenen, J.; Price, R.
(114) Jiang, P. B.; Li, X. L.; Gao, W. B.; Wang, X.; Tang, Y.; Lan, K.; 1521
1454 Furfural�A promising platform for lignocellulosic biofuels. Chem-
1455 SusChem 2012, 5 (1), 150−166.
Wang, B.; Li, R. Highly selective hydrogenation of α,β-unsaturated 1522
carbonyl compounds over supported Co nanoparticles. Catal. Commun. 1523
1457 furfurals to drop-in biofuels: an overview. ACS Sustain. Chem. Eng.
1458 2015, 3 (7), 1263−1277.
(115) Chen, H.; Ruan, H. H.; Lu, X. L.; Fu, J.; Langrish, T.; Lu, X. Y. 1525
Efficient catalytic transfer hydrogenation of furfural to furfuryl alcohol 1526
1459
(98) Addepally, U.; Thulluri, C. Recent progress in production of fuel
in near-critical isopropanol over Cu/MgO-Al2O3 catalyst. Mol. Catal. 1527
1461 catalytic routes. Fuel 2015, 159, 935−942.
(116) Wu, J.; Zhang, X.; Chen, Q.; Chen, L.; Liu, Q.; Wang, C.; Ma, L. 1529
1462
(99) Guzmán, I.; Heras, A.; Güemez, M. B.; Iriondo, A.; Cambra, J. F.;
One-pot hydrogenation of furfural into tetrahydrofurfuryl alcohol 1530
1463 Requies, J. Levulinic acid production using solid-acid catalysis. Ind. Eng.
under ambient conditions over PtNi alloy catalyst. Energy Fuels 2020, 1531
1464 Chem. Res. 2016, 55 (18), 5139−5144.
34 (2), 2178−2184.
1532
1465
(100) Audemar, M.; Ciotonea, C.; De Oliveira Vigier, K.; Royer, S.;
(117) Li, Z. X.; Wei, X. Y.; Yang, Z.; Zhang, M.; Niu, S.; Meng, X. L.; 1533
1466 Ungureanu, A.; Dragoi, B.; Dumitriu, E.; Jerome, F. Selective
1467 Hydrogenation of furfural to furfuryl alcohol in the presence of a
Li, J.; Zong, Z.-M. Highly selective hydrogenation of furfural to furan-2- 1534
ylmethanol over a Cu/C derived from copper-organic frameworks. 1535
1468 recyclable cobalt/SBA-15 catalyst. ChemSusChem 2015, 8 (11), 1885−
Catal. Commun. 2019, 129, 105679.
1536
1469 1891.
(118)
Maderuelo-Solera, R.; López-Asensio, R.; Cecilia, J. A.; 1537
1470
(101) Yuan, Q. Q.; Zhang, D. M.; Haandel, L. V.; Ye, F. Y.; Xue, T.;
Jiménez-Gómez, C. P.; García- Sancho, C.; Moreno-Tost, R.; 1538
1471 Hensen, E. J. M.; Guan, Y. J. Selective liquid phase hydrogenation of
1472 furfural to furfuryl alcohol by Ru/Zr-MOFs. J. Mol. Catal. A-Chem.
Maireles-Torres, P. Catalytic transfer hydrogenation of furfural to 1539
furfuryl alcohol over calcined MgFe hydrotalcites. Appl. Clay Sci. 2019, 1540
1473 2015, 406, 58−64.
183, 105351.
1541
1474
(102) Fulajtárova, K.; Soták, T.; Hronec, M.; Vávra, I.; Dobrocka, E.;
(119) Li, Z. X.; Wei, X. Y.; Yang, Z.; Zhang, M.; Meng, X. L.; Niu, S.; 1542
1475 Omastová, M. Aqueous phase hydrogenation of furfural to furfuryl
1476 alcohol over Pd-Cu catalysts. Appl. Catal. A-Gen. 2015, 502, 78−85.
Zhang, D.; Zong, Z. M. Highly selective hydrogenation of furfural to 1543
furan-2-ylmethanol over ZIF-67-templated magnetic Cu-Co/C. Catal. 1544
1477
(103) Wang, F.; Zhang, Z. H. Catalytic transfer hydrogenation of
Lett. 2020, 150 (1), 178−184.
1545
1478 furfural into furfuryl alcohol over magnetic γ-Fe2O3@HAP catalyst.
1479 ACS Sustainable Chem. Eng. 2017, 5 (1), 942−947.
(120) Xu, L.; Nie, R.; Lyu, X.; Wang, J.; Lu, X. Selective hydrogenation 1546
of furfural to furfuryl alcohol without external hydrogen over N-doped 1547
1480
(104) Gong, W. B.; Chen, C.; Zhang, Y.; Zhou, H. J.; Wang, H. M.;
carbon confifined Co catalysts. Fuel Process. Technol. 2020, 197, 1548
1482 of furfuryl alcohol from H2-hydrogenation/transfer hydrogenation of 1483 furfural using sulfonate group modified Cu catalyst. ACS Sustainable 1484 Chem. Eng. 2017, 5 (3), 2172−2180.
(121) Li, Z. X.; Wei, X. Y.; Liu, G. H.; Meng, X. L.; Yang, Z.; Niu, S.; 1550 Zhang, D.; Gao, H. S.; Ma, Z. H.; Zong, Z. M. Highly selective 1551 hydrogenation of furfural and levulinic acid over Ni0.09Zn/NC600 1552
1485
(105) Dohade, M. G.; Dhepe, P. L. Efficient hydrogenation of
derived from ZIFW-8. Mol. Catal. 2020, 480, 110651.
1553
1486 concentrated aqueous furfural solutions in to furfuryl alcohol under 1487 ambient conditions in presence of PtCo bimetallic catalyst. Green Chem. 1488 2017, 19, 1144−1154.
(122) Li, Z. X.; Wei, X. Y.; Yang, Z.; Li, J.; Yan, W. W.; Bie, L. L.; 1554 Zhang, Y. Y.; Zong, Z. M.; et al. Selective hydrogenation of bio-based 1555 furfural over Co-based catalysts derived from zeolitic imidazolate frame 1556
1489
(106) Wu, J.; Gao, G.; Li, J. L.; Sun, P.; Long, X. D.; Li, F. W. Efficient
materials. Mol. Catal. 2020, 492, 111007.
1557
1490 and versatile CuNi alloy nanocatalysts for the highly selective
(123)
Borah, P.; Yamashita, Y.; Kobayashi, S. Solid superbase- 1558
1491 hydrogenation of furfural. Appl. Catal., B 2017, 203, 227−236.
catalyzed stereoselective 1,4-addition reactions of simple amides in 1559
1560 batch and continuous-flow systems. Adv. Synth. Catal. 2019, 361,
1561 3807−3812.
(142) Wang, D.; Wang, Y.; Li, X.; Chen, L.; Li, G.; Li, X. Lignin 1629
Valorization: A novel in situ catalytic hydrogenolysis method in alkaline 1630
1562
(124) Li, M.; Wang, S.; Li, F.; Zhou, L.; Lei, L. Iodine-mediated
aqueous solution. Energy Fuels 2018, 32 (7), 7643−7651.
1631
1563 photo-controlled atom transfer radical polymerization (photo-ATRP)
1564 and block polymerization combined with ring-opening polymerization
(143) Reynoso, A. J.; Ayastuy, J. L.; Iriarte-Velasco, U.; Gutiérrez- 1632
Ortiz, M. A. Cobalt aluminate spinel-derived catalysts for glycerol 1633
1565 (ROP) via a superbase. Polym. Chem. 2020, 11, 6591−6598.
aqueous phase reforming. Appl. Catal. B Environ. 2018, 239, 86−101.
1634
1566
(125) Shigeno, M.; Hayashi, K.; Korenaga, T.; Nozawa-Kumada, K.;
(144) Morales-Marín, A.; Ayastuy, J. L.; Iriarte-Velasco, U.; Gutiérrez- 1635
1567 Kondo, Y. Organic superbase t-bu-P4-catalyzed demethylations of
1568 methoxyarenes. Org. Chem. Front. 2022, 9, 3656−3663.
Ortiz, M. A. Nickel aluminate spinel-derived catalysts for the aqueous 1636
phase reforming of glycerol: Effect of reduction temperature. Appl. 1637
1569
(126) Zhang, F. C.; Zhang, Q. X.; Liu, P.; Zhang, K.; Ji, K. M.; Zhang,
Catal. B Environ. 2019, 244, 931−945.
1638
1570 J. L.; Zhao, L.; Song, Q. W. Selective synthesis of dimethyl carbonate via
1571 the coupling reaction of CO2 and alcohols by the synergistic catalysis of
1572 silver sulfadiazine and superbase. J. Fuel Chem. Technol. 2023, 51, 304−
1573 312.
(145) Zhang, X. Q.; Kang, Y. H.; Gao, J.; Xiong, L.; Gao, Y.; Chen, T.; 1639 Liu, G. H.; Wang, A. M.; Wei, X. Y.; Zong, Z. M.; Bai, H. C. Effective 1640 hydroconversion of heteroatom-containing organic species from the 1641 extraction of low-temperature coal tar to cycloalkanes over a Y/Beta 1642
1574
(127) Wang, Z.; Wang, Z.; Huang, X.; Yang, D.; Wu, C.; Chen, J. Deep
composite zeolite supported nickel nanoparticles. Fuel 2022, 321, 1643
1576 liquids and ethylene glycol for CO2 capture. Chem. Commun. 2022, 58,
1577 2160−2163.
(146) Liu, G. H.; Bai, J. J.; Lu, K. L.; Kan, H.; Zhang, X. Q.; Kang, Y. 1645
H.; Lu, C. Y.; Li, Z. K.; Bai, H. C.; Wei, X. Y. Integrated catalytic 1646
1578
(128) Ishaq, M.; Gilani, M. A.; Arshad, I.; Bilad, M. R.; Ahmad, F.;
hydroconversion of three crop stalks to valuable oxygenated organic 1647
1579 Khan, A. L. Synergy of high permeability and selectivity of superbase/
chemicals. Fuel 2022, 322, 124149.
1648
1580 choline chloride/urea solution impregnated membranes for CO2
1581 capture. Carbon Capture Sci. Technol. 2021, 1, 100019.
(147) Li, L. Catalytic hydroconversion of Runbei lignite and Shaerhu 1649
subbituminous coal. Doctoral dissertation of China University of Mining 1650
1582
(129) Li, W. T.; Wei, X. Y.; Li, X. K.; Liu, X. X.; Li, Z. K.; Zong, Z. M.
& Technology 2023, Xuzhou, Jiangsu, China.
1651
1583 Catalytic hydroconversion of lignite-related model compounds over
1584 difunctional Ni-Mg2Si/-Al2O3. Fuel 2017, 200, 208−217.
(148) Wei, X. Y.; Zong, Z. M.; Zhao, W.; Ni, Z. H.; Cao, J. P.; Fan, X.; 1652
Zhao, Y. P.; Liu, Z. W.; Peng, Y. L.; Liang, J.; Zhao, X. Y.; Tao, X. Y.; 1653
1585
(130) Liu, Z. Q.; Wei, X. Y.; Liu, F. J.; Liu, G. H.; Zong, Z. M. Catalytic
Kang, Y. H.; Mo, W. L.; Cong, X. S.; Wang, Y. G.; Liu, Z. Q.; Liu, G. H.; 1654
1586 hydroconversion of Yiwu lignite over solid superacid and superbase.
1587 Fuel 2019, 238, 473−482.
Guo, X. H.; Ma, Z. H.; Gao, H. S.; Li, J. H.; Chen, Y. F.; Yan, W. W.; Yin, 1655
F.; Jiang, Z. J.; Yu, X. K. Basic research and technology development for 1656
1588
(131) Liu, X. X.; Zong, Z. M.; Li, W. T.; Li, X.; Li, Z. K.; Wang, S. K.;
the separation of condensed aromatics from high-temperature coal tar. 1657
1589 Wei, X. Y. A recyclable and highly active magnetic solid superbase for
Acta Petrolei Sinica (Petroleum Processing Section) 2022, 38 (3), 500− 1658
|
1591 2017, 159, 396−403. 1592 (132) Zhang, M.; Wei, X. Y.; Yang, Z.; Teng, D. G.; Xue, Y.; Meng, D. 1593 W.; Zong, Z. M. Catalytic hydroconversion of the extraction residue 1594 from Naomaohu lignite over an active and separable magnetic solid 1595 superbase. Fuel 2018, 226, 410−416. 1596 (133) Xu, M. L.; Wei, X. Y.; Liu, G. H.; Li, F. H.; Zhao, Y. P.; Liu, F. J.; 1597 Li, S.; Zong, Z. M.; Fan, X.; Cao, J. P.; Zhao, W.; Ma, F. Y.; Liu, J. M. 1598 Directional catalytic hydroconversion of oxybis(methylene)dibenzene 1599 and extract from Piliqing subbituminous coal over a magnetic 1600 difunctional solid superbase. ChemistrySelect 2020, 5, 1130−1134. 1601 (134) Xu, M. L.; Wei, X. Y.; Wu, Q. C.; Zhao, Y. P.; Li, F. H.; Liu, G. 1602 H.; Liu, F. J.; Zong, Z. M.; Zhang, M.; Li, S.; Fan, X.; Zhao, W.; Ma, F. 1603 Y.; Mo, W. L.; Liu, J. M. Enhanced hydrogenation of aromatic rings and 1604 hydrocracking of > Car-O- bridged bonds in the extraction residue from |
|
(149) Wei, X. Y. Directional conversion of carbon resources to 1660 advanced materials. Symposium on integrated production technology for 1661 the deep processing of new chemical materials and their downstream 1662 products; Ordos, China, 2023. 1663 (150) Shi, D. L.; Wei, X. Y.; Fan, X.; Zong, Z. M.; Chen, B.; Zhao, Y. 1664 P.; Wang, Y. G.; Cao, J. P. Characterizations of the extracts from Geting 1665 bituminous coal by spectrometries. Energy Fuels 2013, 27 (7), 3709− 1666 3717. 1667 (151) Gao, Y.; Wei, X. Y.; Li, Y. J.; Bai, J. J.; Kang, Y. H.; Liu, G. H.; 1668 Ma, X. R.; Li, X.; Lu, C. Y.; Bai, H. C.; Zong, Z. M. Investigation on the 1669 composition of soluble portions from the extraction residue of 1670 Hanglaiwan subbituminous coal by thermal dissolution and alkanol- 1671 yses. Fuel 2021, 306, 121747. 1672 (152) Liu, Z. W.; Zong, Z. M.; Li, J. N.; Chen, C. F.; Jiang, H.; Peng, Y. 1673 |
|
1605 Piliqing subbituminous coal over a magnetic difunctional solid |
|
L.; Xue, J. Q.; Yang, X. L.; Zheng, Y. X.; Zhou, X.; Xie, R. L.; Wei, X. Y. 1674 |
|
1606 superbase. J. Anal. Appl. Pyrolysis 2020, 146, 104695. |
|
Isolation and identification of two bis(2-ethylheptyl) benzenedicarbox- 1675 |
|
1607 (135) Liu, F. J.; Guo, J. P.; Liu, G. H.; Bie, L. L.; Zhao, Y. P.; Huang, Z. |
|
ylates from Lingwu coal. Energy Fuels 2009, 23 (1), 588−590. 1676 |
|
1608 X.; Wei, X. Y. Effect of temperature on catalytic hydrocracking of |
|
(153) Zong, Y.; Zong, Z. M.; Ding, M. J.; Zhou, L.; Huang, Y. G.; 1677 |
|
1609 Xiaolongtan lignite over a mesoporous silica-coated Fe3O4 supported 1610 magnetic solid base for producing aromatics. J. Energy Inst. 2021, 94, |
|
Zheng, Y. X.; Jin, X.; Ma, Y. M.; Wei, X. Y. Separation and analysis of 1678 organic compounds in an Erdos coal. Fuel 2009, 88 (3), 469−474. 1679 |
|
1611 352−359. |
|
(154) Liu, Z. W.; Wei, X. Y.; Zong, Z. M.; Li, J. N.; Xue, J. Q.; Chen, X. 1680 |
|
1612 (136) Mathews, J. P.; Chaffee, A. L. The molecular representations of |
|
F.; Chen, F. J. Isolation and identification of methyl alkanoates from 1681 |
|
1613 coal - A review. Fuel 2012, 96, 1−14. |
|
Lingwu coal. Energy Fuels 2010, 24 (4), 2784−2786. 1682 |
|
1614 (137) Liu, F. J.; Wei, X. Y.; Fan, M. H.; Zong, Z. M. Separation and |
|
(155) Cong, X. S.; Zong, Z. M.; Zhou, Y.; Li, M.; Wang, W. L.; Li, F. 1683 |
|
1615 structural characterization of the value-added chemicals from mild |
|
G.; Zhou, J.; Fan, X.; Zhao, Y. P.; Wei, X. Y. Isolation and identification 1684 |
|
1616 degradation of lignites: A review. Appl. Energy 2016, 170, 415−436. |
|
of 3-ethyl-8-methyl-2,3-dihydro-1H-cyclopenta[a]- chrysene from 1685 |
|
1617 (138) Liu, G. H.; Li, Y. J.; Bai, J. J.; Gao, Y.; Kang, Y. H.; Wang, A. M.; |
|
Shengli lignite. Energy Fuels 2014, 28 (10), 6694−6697. 1686 |
|
1618 Lu, C. Y.; Bai, H. C.; Zong, Z. M.; Wei, X. Y. Advances in mild |
|
(156) Cong, X. S.; Zong, Z. M.; Wei, Z. H.; Li, Y.; Fan, X.; Zhou, Y.; Li, 1687 |
|
1619 degradation and directional upgrading of lignites: From feature |
|
M.; Zhao, Y. P.; Wei, X. Y. Enrichment and identification of arylhopanes 1688 |
|
1620 identification to value-added utilization. J. Anal. Appl. Pyrolysis 2022, |
|
from Shengli lignite. Energy Fuels 2014, 28 (11), 6745−6748. 1689 |
|
1621 163, 105477. |
|
(157) Cong, X. S.; Zong, Z. M.; Li, M.; Zhou, Y.; Gao, S. Q.; Wei, X. Y. 1690 |
|
1622 (139) Benson, S. W. Bond energies. J. Chem. Educ. 1965, 42, 502−508. |
|
Isolation and identification of two novel condensed aromatic lactones 1691 |
|
1623 (140) Kerr, J. A. Bond dissociation energies by kinetic methods. Chem. |
|
from Zhundong subbituminous coal. Energy Fuels 2014, 28 (12), 1692 |
|
1624 Rev. 1966, 66, 465−500. |
|
7394−7397. 1693 |
|
1625 (141) Liu, G. H.; Qi, Y. C.; Wang, H.; Wang, A. M.; Kang, Y. H.; Gao, |
|
(158) Cong, X. S.; Zong, Z. M.; Zhou, Y.; Li, M.; Zhao, Y. P.; Fan, X.; 1694 |
|
1626 Y.; Bai, N.; Li, Z. K.; Wei, X. Y. Synergistic transfer of active hydrogen in |
|
Wei, X. Y. Enrichment and analysis of long-chain normal alkanals from 1695 |
|
1627 the catalytic hydroconversion of lignite-derived aryl ethers to cyclanes: |
|
Zhundong subbituminous coal. J. Fuel Chem. Technol. 2014, 42 (3), 1696 |
|
1628 A combined experimental and DFT study. Fuel 2023, 351, 129034. |
|
257−261. 1697 |
|
|
S |
1699 X.; Zhou, Y.; Wei, X. Y. Enrichment and identification of cyclized
1700 hopanoids from Shengli lignite. Fuel Process. Technol. 2015, 134, 399−
1701 403.
1702
(160) Liu, G. H.; Zong, Z. M.; Liu, F. J.; Meng, X. L.; Zhang, Y. Y.;
1703 Wang, S. K.; Li, S.; Zhu, C.; Wei, X. Y.; Ma, F. Y.; Liu, J. M. Deep
1704 hydroconversion of ethanol-soluble portion from the ethanolsis of 1705 Dahuangshan lignite to clean liquid fuel over a mordenite supported 1706 nickel catalyst. J. Anal. Appl. Pyrolysis 2019, 139, 13−21.
1707
(161) Guan, Y. S.; Zhao, W.; Zhao, C.; Wang, D. K.; Ao, L.; Liu, K. S.;
1708 Liu, H. M.; Chen, S. L.; Fan, X.; Wei, X. Y. A novel magnetic solid acid 1709 for specially cleaving the C-O bridged bond in dibenzyl ether. 1710 ChemistrySelect 2018, 3 (41), 11610−11615.
1711
(162) Zhao, M. X.; Wei, X. Y.; Qu, M.; Li, Z. K.; Liu, J.; Li, Y.; Kong, J.;
1712 Zhao, W.; Zong, Z. M. Hydrocracking of benzyloxybenzene as a lignite- 1713 related model compound over a novel solid acid. Fuel Process. Technol. 1714 2016, 146, 110−115.